Mineral Absorption Modulators: Mechanisms, Methodologies, and Clinical Translation for Drug Development
This comprehensive review synthesizes current scientific knowledge on the complex factors governing mineral bioavailability, with a specific focus on applications in pharmaceutical and therapeutic development.
Mineral Absorption Modulators: Mechanisms, Methodologies, and Clinical Translation for Drug Development
Abstract
This comprehensive review synthesizes current scientific knowledge on the complex factors governing mineral bioavailability, with a specific focus on applications in pharmaceutical and therapeutic development. The article explores foundational absorption mechanisms, including paracellular and transcellular pathways, and details the dietary compounds and pharmaceutical agents that significantly inhibit or enhance mineral uptake. It critically evaluates advanced methodologies for assessing bioavailability, from in vitro digestion models to predictive algorithms and clinical balance studies. The content further examines cutting-edge optimization strategies, including engineered probiotics, permeation-enhancing technologies, and novel fortification approaches. Designed for researchers and drug development professionals, this review provides a robust framework for developing enhanced mineral delivery systems and addressing mineral deficiencies through targeted biomedical interventions.
Fundamental Mechanisms of Mineral Absorption and Bioavailability Barriers
Intestinal mineral absorption is a critical process governed by two primary pathways: the paracellular pathway, which facilitates passive diffusion between epithelial cells, and the transcellular pathway, which involves active transport across the cell membrane. These mechanisms collectively maintain mineral homeostasis, essential for numerous physiological functions including bone mineralization, nerve conduction, muscle contraction, and enzyme function [1] [2]. The efficiency of these pathways is influenced by a complex interplay of dietary factors, gut microbiota, host physiology, and regulatory signaling molecules [3] [4]. Within the context of mineral absorption research, understanding these core mechanisms provides the foundational knowledge required to identify and characterize specific inhibitors and enhancers that can modulate bioavailability. This whitepaper provides a comprehensive technical overview of these mechanisms, detailing the key transporters, regulatory systems, and experimental approaches relevant for researchers and drug development professionals working to optimize mineral absorption.
Paracellular Pathway
The paracellular pathway allows for the passive movement of minerals and other solutes through the intercellular space between adjacent epithelial cells. This route is governed by the tight junction (TJ) complex, a specialized structure that forms a selective seal at the apical region of epithelial cells [5]. The permeability of this pathway is determined by the specific protein composition of the TJs.
Molecular Composition and Regulation
The TJ complex is composed of several key proteins, including claudins, occludin, and zonula occludens (ZO) proteins. Among these, the claudin family plays a particularly crucial role in defining the charge and size selectivity of the paracellular pore [5]. Specifically, claudin-2 forms cation-selective channels, thereby creating a bias for the absorption of positively charged ions, such as calcium, magnesium, and sodium [5] [6]. The expression and cellular distribution of claudin-2 can be dynamically regulated. Research has shown that the peptide PIP 640 can increase the phosphorylation of myosin light chain (MLC-pS19), leading to an increase in claudin-2 expression and its shift from the nucleus to the cellular membrane. This results in a transient enhancement of paracellular permeability, particularly for cationic solutes the size of small therapeutic peptides [5].
This pathway is physiologically distinct from the barrier disruption caused by pro-inflammatory cytokines like TNF-α and IFN-γ. While cytokine exposure also increases MLC phosphorylation, it typically leads to a more non-selective increase in permeability, including for larger molecules, and is associated with a decrease in occludin expression [5]. The paracellular route is most effective for mineral transport when the luminal concentration of a mineral is higher than its concentration in the bloodstream, creating a favorable concentration gradient [6].
Table 1: Key Proteins in the Paracellular Pathway
| Protein Name | Function | Regulation & Notes |
|---|---|---|
| Claudin-2 | Forms cation-selective paracellular channels. | Increased by PIP 640; creates bias for positively charged minerals [5]. |
| Occludin | Contributes to TJ barrier integrity. | Decreased by pro-inflammatory cytokines (TNF-α, IFN-γ) [5]. |
| Zonula Occludens (ZO-1, ZO-2, ZO-3) | Scaffold proteins linking transmembrane TJ proteins to the actin cytoskeleton. | Essential for TJ assembly and function [6]. |
| Tricellulin | Seals TJs at the convergence of three cells. | Decreased by sodium caprate, enhancing permeability at tricellular locations [5]. |
Transcellular Pathway
The transcellular pathway is an active, energy-dependent process that transports minerals directly through the intestinal epithelial cell. This pathway is essential for the absorption of minerals when their luminal concentration is low, requiring movement against a concentration gradient. It involves a three-step process: entry across the apical membrane, intracellular trafficking through the cytosol, and exit across the basolateral membrane into the circulation [6].
Key Transporters and Channels
Transcellular transport is mediated by a suite of specialized transporters, channels, and pumps that are often specific to particular minerals.
- Calcium: Entry across the apical membrane is primarily mediated by Transient Receptor Potential Vanilloid (TRPV) channels, such as TRPV6. Intracellular calcium is then shuttled across the cell by calcium-binding proteins like calbindin-D and actively extruded at the basolateral membrane by the Plasma Membrane Ca²⁺ ATPase (PMCA) and the Sodium-Calcium Exchanger (NCX1) [6].
- Phosphorus: Inorganic phosphorus is absorbed via the sodium-dependent phosphate transporter NaPi-IIb (SLC34A2) [6].
- Other Minerals: Transporters for other minerals include ZIP4 for zinc, and DMT1 for non-heme iron, though the latter can also be influenced by the gut microbiota [3].
The expression and activity of many of these transcellular transporters are under strict hormonal control. The hormonally active form of vitamin D, 1α,25-dihydroxyvitamin D₃ [1α,25(OH)₂D₃], is a primary regulator, upregulating the expression of TRPV6 and calbindin-D to enhance calcium absorption [6]. Other hormones, including Parathyroid Hormone (PTH) and Fibroblast Growth Factor 23 (FGF23), also interact with this system to maintain systemic mineral homeostasis [6].
Table 2: Major Transcellular Transporters for Minerals
| Mineral | Apical Influx Transporter | Cytosolic Transport | Basolateral Efflux Transporter |
|---|---|---|---|
| Calcium (Ca²⁺) | TRPV6, TRPV2, TRPM7 [6] | Calbindin-D [6] | PMCA2, PMCA4, NCX1 [6] |
| Inorganic Phosphorus (Pᵢ) | NaPi-IIb (SLC34A2) [6] | - | PiT-1, PiT-2 [6] |
| Zinc (Zn²⁺) | ZIP4 [3] | - | ZnT-1 [3] |
| Iron (Fe) | DMT1 [3] | - | Ferroportin [3] |
Key Regulatory Systems
Endocrine Regulation
Systemic mineral homeostasis is predominantly regulated by a hormonal axis involving vitamin D, parathyroid hormone (PTH), and fibroblast growth factor 23 (FGF23).
- Vitamin D Metabolism: The active form, 1α,25(OH)₂D₃, is synthesized in the kidney via the enzyme 1α-hydroxylase (CYP27B1). It acts by binding to the Vitamin D Receptor (VDR) in the nucleus of enterocytes, forming a heterodimeric complex with Retinoid X Receptors (RXR). This complex acts as a transcription factor to upregulate the expression of transcellular transport proteins like TRPV6 and calbindin-D [6].
- Parathyroid Hormone (PTH): Secreted in response to low blood calcium levels, PTH binds to its receptor PTH1R in the kidney to stimulate the production of 1α,25(OH)₂D₃, thereby indirectly enhancing intestinal calcium absorption [6].
- Fibroblast Growth Factor 23 (FGF23): This bone-derived hormone is released in response to high phosphate levels. It signals through FGFR receptors along with the co-receptor klotho to exert phosphaturic effects and suppress the formation of 1α,25(OH)₂D₃, thus reducing intestinal phosphate absorption [6].
The Gut Microbiota and Mineral Absorption
The gut microbiota plays a significant role as an enhancer of mineral bioavailability. Beneficial bacteria, particularly from the Lactobacillus and Bifidobacterium families, can improve mineral absorption through several mechanisms [3] [4].
- Phytate Degradation: Many plant-based foods contain phytic acid, a potent antinutritional factor that chelates minerals and impedes their absorption. Certain probiotic strains produce the enzyme phytase, which breaks down phytic acid, releasing bound minerals such as calcium, magnesium, and zinc and making them available for absorption [3].
- Production of SCFAs: Microbial fermentation of dietary fiber produces short-chain fatty acids (SCFAs) like acetate, propionate, and butyrate. SCFAs help lower the intestinal pH, which can increase the solubility of certain minerals, and may also influence the expression of tight junction proteins, potentially modulating paracellular permeability [4].
- Modulation of Transporters: Microbial metabolites can influence the expression of host mineral transporters, although the precise mechanisms are still under investigation [3].
Experimental Models and Methodologies
In Vitro Models
Caco-2 cell monolayers are a cornerstone of intestinal absorption research. Derived from human colon adenocarcinoma, these cells spontaneously differentiate into enterocyte-like cells when cultured on permeable supports, forming polarized monolayers with well-developed tight junctions and expressing many relevant transporters [5] [7].
- Protocol: Transport Studies: Caco-2 cells are cultured on 1.2 cm², 0.4 μm pore polyester membranes in 12-well Transwell plates. Cells are seeded at a density of 7 × 10⁴ cells/well and cultured for approximately 2 weeks until fully polarized, as indicated by a trans-epithelial electrical resistance (TEER) value of > ~350 Ω·cm². Test compounds (e.g., PIP 640) are applied to the apical compartment. Transport is assessed by measuring the apical-to-basolateral (AB) flux of marker molecules (e.g., 4-kDa dextran) over time. Sampling from the basolateral compartment at regular intervals allows for the calculation of apparent permeability (Pₐₚₚ) [5].
- Protocol: Gene/Protein Expression Analysis: Following treatments, total cell lysates, nuclear, or membrane fractions of the monolayers are prepared. Proteins are separated by SDS-PAGE, transferred to PVDF membranes, and probed with specific primary antibodies against proteins of interest (e.g., claudin-2, occludin, MLC-pS19). Actin is typically used as a loading control [5].
- Bioavailability Assessment: The Caco-2 model is widely used to assess iron bioavailability by measuring ferritin formation. After exposure to digested food samples (e.g., from processed lentils), cellular ferritin levels are quantified using an ELISA kit. Ferritin formation is a sensitive indicator of cellular iron uptake and utilization [7].
In Vivo Models
Rodent models (rats and mice) are commonly used for validating findings from in vitro studies and investigating complex systemic regulation.
- Protocol: Luminal Injection Studies: To study the acute effects of an absorption enhancer in a specific intestinal segment, a surgical approach can be used. For example, a peptide like PIP 640 can be injected directly into a ligated segment of the jejunum in anesthetized rats. After a designated period, the segment is collected for analysis of TJ protein localization (via immunohistochemistry) and measurement of therapeutic peptide transport into systemic circulation or adjacent tissues [5].
- The Gallus gallus (Chicken) Model: This model is particularly useful for longitudinal studies on mineral metabolism, especially concerning bone health and development. Its advantage includes the ease of administering controlled diets and assessing functional outcomes like bone mineralization [7].
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for Investigating Intestinal Mineral Uptake
| Reagent / Tool | Function/Application | Example Use Case |
|---|---|---|
| Caco-2 Cell Line | Human intestinal epithelial cell model for permeability and transport studies. | Cultured on Transwell inserts to form polarized monolayers for testing solute flux and TEER [5] [7]. |
| PIP 640 Peptide | A cell-penetrating peptide that inhibits myosin light chain phosphatase. | Apical application to increase MLC-pS19 and claudin-2 levels, enhancing cation-selective paracellular permeability [5]. |
| Transwell Plates | Permeable supports for culturing polarized cell monolayers. | Used with Caco-2 cells to create apical and basolateral compartments for transport assays [5]. |
| Cytokine Cocktails (TNF-α/IFN-γ) | Pro-inflammatory cytokines used to model inflammatory barrier disruption. | Basolateral treatment to induce a non-selective leak pathway, decreasing occludin expression [5]. |
| Specific Antibodies | For detection and localization of key proteins via Western blot or immunofluorescence. | Antibodies against claudin-2, occludin, ZO-1, MLC-pS19, and TRPV6 are essential for mechanistic studies [5] [6]. |
| In Vitro Digestion Model | Simulates human gastric and intestinal digestion. | Used to process food samples (e.g., lentil flour) prior to assessing mineral bioaccessibility and bioavailability in Caco-2 assays [7]. |
| TEER Measurement System | Measures Trans-Epithelial Electrical Resistance to monitor monolayer integrity. | Used to validate the quality of Caco-2 monolayers before and during experiments [5]. |
The intricate interplay between the paracellular and transcellular pathways forms the foundation of intestinal mineral uptake. The paracellular route, governed by tight junction dynamics, offers a passive, charge-selective route, while the transcellular pathway provides an active, regulated mechanism for specific minerals. This core understanding is vital for advancing research into inhibitors and enhancers of mineral absorption. Current research is increasingly revealing the critical modulatory roles of the gut microbiota and dietary components, offering novel avenues for intervention. The experimental frameworks and tools outlined in this whitepaper, ranging from well-established cell models to sophisticated in vivo protocols, provide researchers with a robust methodology for dissecting these mechanisms. Future work aimed at precisely targeting these pathways holds significant promise for improving mineral bioavailability in both nutritional and therapeutic contexts, potentially addressing widespread mineral deficiencies and related health burdens.
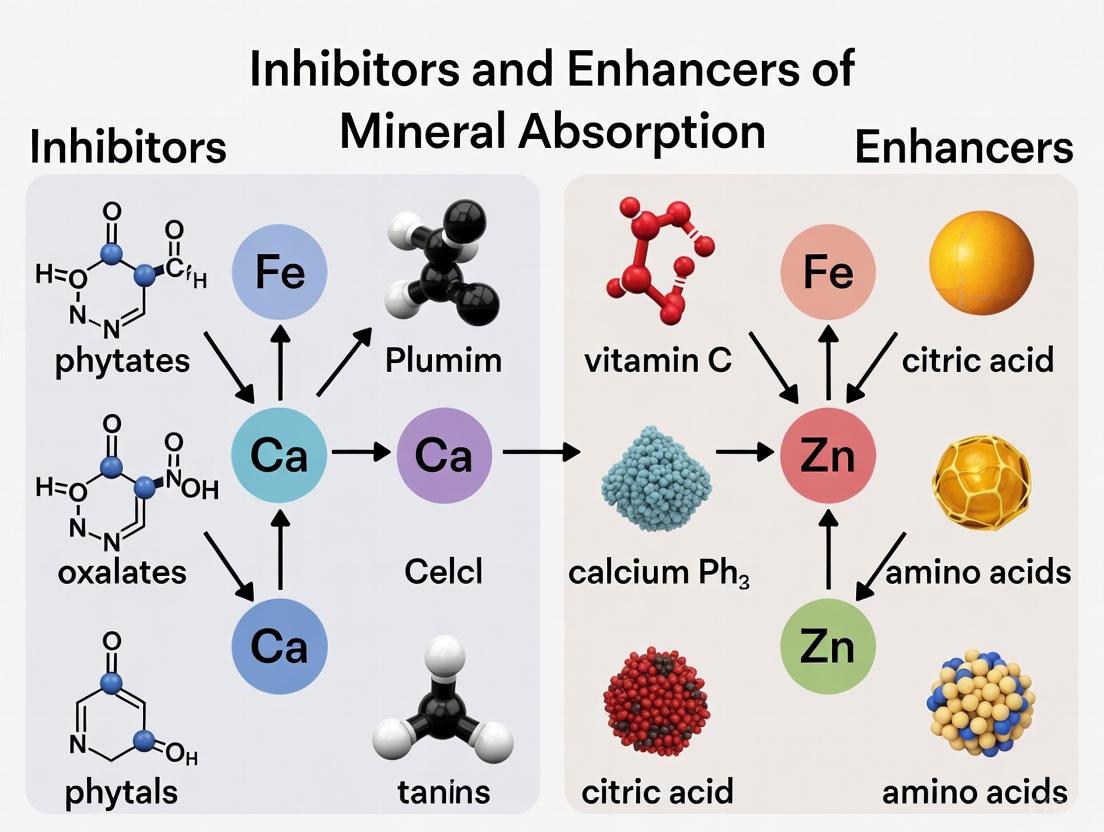
Dietary antinutrients are naturally occurring compounds in plant-based foods that can interfere with the absorption of essential minerals in the human gastrointestinal tract. Within the broader context of research on inhibitors and enhancers of mineral absorption, this review provides a comprehensive analysis of three prominent antinutrients: phytates, oxalates, and tannins. These compounds form complexes with minerals such as iron, zinc, and calcium, reducing their bioavailability and potentially contributing to micronutrient deficiencies. Understanding their mechanisms of action, factors influencing their activity, and methodologies to mitigate their effects is crucial for addressing global malnutrition challenges and formulating strategies to enhance mineral bioavailability from plant-based foods, which constitute a significant portion of diets worldwide, particularly in developing countries [8] [9].
Mechanistic Actions of Major Dietary Antinutrients
Phytates (Phytic Acid)
Phytic acid (myo-inositol 1,2,3,4,5,6-hexakis dihydrogen phosphate or IP6) serves as the primary storage form of phosphorus in cereals, legumes, oil seeds, and nuts, comprising 1% to 5% by weight of these materials [9]. Its strong chelating property enables it to bind minerals such as iron, zinc, calcium, and magnesium in the gastrointestinal tract, forming insoluble complexes that are poorly absorbed by monogastric animals, including humans, who lack sufficient levels of the phytate-degrading enzyme phytase [9]. This binding results in significantly reduced bioavailability of these essential minerals, with the limited bioavailability of cereal mineral content offering a substantial nutritional challenge [9]. Beyond its antinutritional effects, phytic acid also exhibits antioxidant properties by chelating transition metals like iron, thereby preventing them from catalyzing Fenton reactions that generate damaging hydroxyl radicals [10]. Research has also revealed potential therapeutic benefits, including antineoplastic effects and protection against vascular calcification in patients with kidney disease [8] [10].
Oxalates
Oxalates, or oxalic acid, are small organic acids that plants produce to manage calcium and mineral reserves, regulate water balance, and deter insects [10]. In humans, oxalates act as antinutrients primarily by binding to calcium in the gut, forming insoluble calcium oxalate crystals that prevent calcium absorption [10] [11]. The absorption of calcium from calcium oxalate is substantially lower than from other sources like milk, with fractional calcium absorption averaging 0.100 ± 0.043 when ingested alone compared to 0.358 ± 0.113 from milk [12]. Beyond impairing calcium absorption, oxalates can contribute to kidney stone formation, the most common type being calcium oxalate stones [11]. When excreted in urine, oxalates can bind to calcium, forming crystals that may clump together into painful stones, particularly when urine volume is low [11]. The absorption of oxalate is highly dependent on calcium intake, with one study demonstrating that oxalate absorption decreases linearly from 17% ± 8.3% with a 200 mg daily calcium intake to 2.6% ± 1.5% with a 1200 mg daily calcium intake [13].
Tannins
Tannins represent a broad class of water-soluble polyphenolic compounds, divided into hydrolysable tannins and condensed tannins (proanthocyanidins), with the latter being more commonly consumed [14]. These compounds exert their antinutritional effects primarily by forming complexes with proteins, starch, and digestive enzymes, thereby reducing the nutritional value of foods [15]. Regarding mineral absorption, tannins reduce iron availability before absorption through the formation of insoluble iron-tannin complexes [14]. This interaction particularly affects non-heme iron, the form found in plant foods, which is already less bioavailable than heme iron from animal sources [11]. The effect of tannins on iron status presents a complex picture, with dissonance observed between single-meal studies that generally support reductions in iron bioavailability and long-term studies that often show minimal changes in iron status, suggesting possible adaptive mechanisms [14]. Interestingly, tannins also possess significant antioxidant and anticancer actions, creating a dichotomy between their potential detrimental and beneficial health properties [14] [15].
Quantitative Analysis of Antinutrient Effects
Mineral Binding Capacities and Absorption Inhibition
Table 1: Quantitative Effects of Antinutrients on Mineral Absorption
| Antinutrient | Minerals Affected | Reduction in Absorption | Key Quantitative Findings |
|---|---|---|---|
| Phytates | Iron, Zinc, Calcium, Magnesium | Varies by mineral and food matrix | In unrefined cereals, mineral bioavailability reduced to 5%-15% [9]. Inhibition of non-heme iron absorption varied from 1% to 23% in vegetarians [11]. |
| Oxalates | Calcium | Significant reduction | Calcium absorption from oxalate: 0.100 ± 0.043 vs. 0.358 ± 0.113 from milk [12]. Oxalate absorption decreases from 17% ± 8.3% to 2.6% ± 1.5% as calcium intake increases from 200 to 1200 mg/day [13]. |
| Tannins | Iron (non-heme) | Highly variable | One cup of tea contains 25-80 mg tannins; 3 cups/day (75-240 mg tannins) can inhibit iron absorption in single-meal studies [14]. Long-term effects on iron status less pronounced than single-meal effects [14]. |
Table 2: Antinutrient Content in Common Food Sources
| Food Category | Specific Food | Antinutrient Content | Remarks |
|---|---|---|---|
| Cereals (Phytates) | Wheat bran | 2.1-7.3 g/100 g dw [9] | Higher in bran layers |
| Rice bran | 2.56-8.7 g/100 g dw [9] | Varies by processing | |
| Maize germ | 6.39 g/100 g dw [9] | Concentrated in germ | |
| Legumes (Phytates) | Soybeans | 1.0-2.22 g/100 g dw [9] | Reduced by processing |
| Lentils | 0.27-1.51 g/100 g dw [9] | Varies by cultivar | |
| Oxalate Sources | Spinach (cooked) | 755 mg/½ cup [16] | Boiling reduces content |
| Almonds | 122 mg/oz (22 nuts) [16] | Among highest in nuts | |
| Beets | 152 mg/cup [16] | Consumed cooked or raw | |
| Tannin Sources | Tea | 25-80 mg/150 mL cup [14] | Varies by type, processing |
Experimental Methodologies for Assessing Antinutrient Effects
Mineral Absorption Studies
The primary methodology for assessing mineral bioavailability involves isotopic labeling techniques, where minerals are intrinsically or extrinsically labeled with stable or radioactive isotopes. In one representative study investigating calcium absorption from oxalate, researchers used intrinsically labeled Ca oxalate and compared absorption with calcium from milk in the same subjects under two conditions: when test substances were ingested in separate meals and when ingested together [12]. This within-subject design allowed for direct comparison of calcium absorption from different sources while controlling for interindividual variability. The fractional absorption of calcium was calculated based on the appearance of the isotopic label in blood, urine, or fecal samples, using mathematical models to determine the absorption efficiency [12]. For iron absorption studies, similar isotopic approaches are employed, often using erythrocyte incorporation of stable iron isotopes as the endpoint measurement [14].
In Vitro Bioaccessibility Models
In vitro gastrointestinal simulation models provide a cost-effective screening tool for assessing mineral bioavailability from different food matrices. These models typically simulate gastric and intestinal digestion phases using standardized enzymes, pH adjustments, and incubation times. The bioaccessible fraction of minerals is then quantified in the digested supernatant using analytical techniques such as atomic absorption spectroscopy or inductively coupled plasma mass spectrometry [9]. One significant advantage of in vitro models is the ability to test multiple factors simultaneously under controlled conditions; however, their physiological relevance is limited compared to human trials [14]. These models are particularly useful for screening processing techniques intended to reduce antinutrient content and improve mineral bioavailability before proceeding to more costly human trials.
Long-Term Intervention and Observational Studies
Long-term studies in animal models and human populations provide critical data on the relationship between antinutrient consumption and mineral status over time. Animal studies allow for direct measurement of tissue mineral concentrations, hemoglobin levels, and other functional endpoints following controlled diets containing specific antinutrients [14] [15]. In human research, prospective cohort studies and randomized controlled trials examine associations between dietary patterns high in antinutrients and health outcomes such as anemia prevalence, bone mineral density, or kidney stone incidence [14] [11]. These long-term studies are essential for contextualizing findings from single-meal absorption studies, as they can account for adaptive mechanisms that may develop over time, such as upregulation of mineral transporters or modifications in gut microbiota composition [14].
Methodologies for Mitigating Antinutrient Effects
Traditional Food Processing Techniques
Traditional food processing methods have evolved across cultures specifically to reduce antinutrient content and improve the nutritional quality of plant-based foods. Soaking grains, legumes, and seeds in water facilitates the leaching of water-soluble antinutrients like phytates and oxalates into the soaking medium, which is then discarded [11] [17]. The effectiveness of soaking can be enhanced by using slightly acidic conditions or adding phytase-rich grains. Fermentation utilizes microorganisms that produce phytase and other enzymes capable of degrading antinutrients [9]. Lactic acid bacteria fermentation has been particularly effective at reducing phytate content in cereals and legumes. Germination or sprouting activates endogenous phytase and other enzymes within the plant material, leading to the degradation of phytates and other antinutrients [9] [17]. Thermal processing methods including boiling, autoclaving, and steaming induce denaturation of heat-labile antinutrients such as lectins and goitrogens, while also facilitating the leaching of oxalates and tannins into cooking water [8] [11]. Boiling for 10-12 minutes has been shown to reduce soluble oxalates by 30-87%, with leafy greens like spinach showing reductions of approximately 85% [11].
Emerging Technologies and Biofortification Approaches
Emerging technologies offer promising approaches for reducing antinutrient content in plant-based foods. Ultrasonication applies high-frequency sound waves to disrupt cell walls and facilitate the release and degradation of antinutrients [17]. Enzymatic treatment using exogenous microbial phytase directly targets phytate degradation and has been successfully applied in industrial processing of plant-based ingredients [9]. Genetic engineering and conventional breeding techniques are being employed to develop low-phytate or low-oxalate crop varieties through biofortification approaches [9]. These strategies aim to reduce antinutrient content while maintaining or enhancing nutritional quality and yield characteristics. Biofortification represents a sustainable approach to addressing micronutrient malnutrition, particularly in developing countries where reliance on plant-based staples is high and access to diverse diets or commercial supplements may be limited [9].
Research Reagents and Methodological Tools
Table 3: Essential Research Reagents for Antinutrient Studies
| Reagent/Tool | Function/Application | Examples/Specifications |
|---|---|---|
| Sodium [(13)C₂]oxalate | Isotopically labeled tracer for oxalate absorption studies | Allows precise quantification of oxalate absorption via mass spectrometry [13]. |
| Intrinsically labeled minerals | Tracing mineral absorption from specific food sources | e.g., labeled Ca, Fe, Zn; requires plant growth in isotopic solutions [12]. |
| Phytase enzymes | Degradation of phytic acid in experimental diets | Microbial sources (fungal, bacterial) with defined activity units; used in dephytinization protocols [9]. |
| In vitro gastrointestinal simulation systems | Simulating human digestion for bioavailability screening | Multi-chamber systems with controlled pH, enzymes, mixing; requires standardized digestive enzymes [14] [9]. |
| Atomic Absorption Spectroscopy (AAS) | Quantification of mineral content in food and biological samples | Requires element-specific lamps and standards; alternative: ICP-MS for higher sensitivity [9]. |
| High-Performance Liquid Chromatography (HPLC) | Separation and quantification of antinutrients | Method development required for different antinutrient classes (phytates, tannins, oxalates) [9] [15]. |
Diagram 1: Research Framework for Antinutrient Studies
Diagram 2: Antinutrient Sources, Targets and Mitigation Approaches
Phytates, oxalates, and tannins represent significant challenges to mineral bioavailability from plant-based foods, yet their effects are modifiable through various processing techniques and dietary strategies. The current evidence base demonstrates that while these compounds can substantially reduce mineral absorption in single-meal studies, their long-term impact on mineral status is modulated by adaptive physiological mechanisms and dietary context. Future research should focus on optimizing traditional processing methods, developing novel reduction techniques, and further elucidating the molecular mechanisms underlying mineral-antinutrient interactions. Additionally, more longitudinal human studies are needed to better understand the adaptation phenomena observed with regular consumption of antinutrient-containing foods. Within the broader context of mineral absorption research, a balanced approach that recognizes both the potential drawbacks and health benefits of foods containing these compounds is essential for developing evidence-based dietary recommendations and addressing global micronutrient deficiencies.
The nutritional adequacy of a mineral is not solely a function of its total dietary intake but is fundamentally determined by its bioavailability—the proportion that is absorbed, transported, and utilized in physiological processes [18]. Mineral-mineral interactions at the sites of absorption and transport represent a critical determinant of bioavailability. These interactions, which can be antagonistic (competitive inhibition) or cooperative (synergistic absorption), are governed by the shared use of transport proteins, the physiological status of the individual, and the overall meal composition [19] [20]. Understanding these dynamics is paramount for developing effective nutritional interventions and therapeutic strategies. This whitepaper, situated within a broader thesis on inhibitors and enhancers of mineral absorption, provides a technical guide to the mechanisms, key players, and experimental assessment of mineral-mineral interactions for a research and drug development audience.
Mechanisms of Mineral Absorption and Interaction
Intestinal Absorption Pathways
The intestinal lumen is the primary arena for mineral interactions. Non-heme iron and several divalent cations are absorbed primarily in the duodenum via a shared, pH-dependent pathway. Ferric iron (Fe³⁺) is first reduced to ferrous iron (Fe²⁺) by duodenal cytochrome B (DCYTB). The divalent metal transporter 1 (DMT1) then facilitates the uptake of Fe²⁺ across the apical membrane of the enterocyte [20]. Critically, DMT1 is not specific to iron; it also transports zinc (Zn²⁺), manganese (Mn²⁺), copper (Cu²⁺), and cobalt (Co²⁺) [19] [20]. This shared gateway is a fundamental point of competitive inhibition.
Upon entering the enterocyte, minerals can be stored (e.g., in ferritin), utilized cellularly, or exported into the systemic circulation. Ferroportin (FPN1) is the sole known exporter for iron, a process aided by the ferroxidase activity of hephaestin or ceruloplasmin (Cp), which oxidizes Fe²⁺ back to Fe³⁺ for binding to transferrin (Trf) in the blood [20]. The hormone hepcidin, produced by the liver, regulates systemic iron homeostasis by binding to FPN1 and inducing its internalization and degradation, thereby controlling iron efflux into the circulation [20].
Systemic Regulation and Homeostasis
Systemic homeostasis of essential trace minerals is maintained through tightly regulated absorption and excretion. Most trace minerals, including zinc, copper, and iron, are primarily regulated at the intestinal level, where absorption is modulated in response to physiological demand and body stores [19]. In contrast, elements like selenium and iodine are primarily regulated at the renal level through excretion [19]. Toxic elements such as cadmium (Cd), lead (Pb), and mercury (Hg) can disrupt this delicate balance by interfering with the absorption, transport, and function of essential minerals, exacerbating deficiencies and contributing to chronic disease pathogenesis [19].
Competitive Inhibition: Mechanisms and Key Antagonists
Competitive inhibition occurs when minerals with similar physicochemical properties (e.g., charge and ionic radius) vie for binding sites on shared absorptive transporters or transport proteins, thereby reducing the uptake or utilization of one another.
The following diagram illustrates the key competitive interactions at the major intestinal absorption sites.
Major Competitive Interactions
Calcium and Iron: High doses of supplemental calcium can inhibit the absorption of both heme and non-heme iron by competitively interacting with DMT1 and other undefined transport pathways. This interaction is dose-dependent and is a significant consideration for individuals at risk of iron deficiency who consume calcium-fortified foods or supplements [20].
Zinc and Copper: High zinc intake (well above the Recommended Dietary Allowance) can induce copper deficiency by competitively inhibiting its absorption in the intestine. Zinc stimulates the synthesis of metallothionein in enterocytes, a protein that has a higher binding affinity for copper than for zinc. The bound copper is then trapped in the enterocyte and lost during intestinal cell sloughing [19].
Toxic and Essential Elements: Toxic heavy metals exploit physiological transport pathways, leading to direct competition. Cadmium (Cd) inhibits iron absorption, while lead (Pb) competitively inhibits iron distribution [20]. Mercury (Hg) binds to selenium (Se), preventing its incorporation into essential antioxidant enzymes like glutathione peroxidase [19].
Table 1: Key Competitive Mineral Interactions and Their Physiological Consequences
| Mineral Pair | Site of Interaction | Mechanism | Physiological Impact |
|---|---|---|---|
| Calcium & Iron | Duodenal Enterocyte | Competitive inhibition at DMT1 and other transport proteins [20]. | Reduced iron absorption, potentially exacerbating iron deficiency [20]. |
| Zinc & Copper | Intestinal Lumen / Enterocyte | Zn induction of metallothionein, which sequesters Cu [19]. | Zn-induced Cu deficiency, leading to hematological and neurological impairments [19]. |
| Iron & Zinc | Duodenal Enterocyte (DMT1) | Competition for shared divalent metal transporter [20]. | Excess Fe can hinder Zn absorption; Transferrin can bind both Fe and Zn [20]. |
| Cadmium & Iron | Duodenal Enterocyte (DMT1) | Cd²⁺ competes with Fe²⁺ for absorption via DMT1 [20]. | Reduced Fe uptake, potentially increasing risk of anemia [19] [20]. |
| Manganese & Iron | Duodenal Enterocyte (DMT1) | Mn²⁺ competes with Fe²⁺ for absorption via DMT1 [20]. | Altered status of both minerals; relevant in parenteral nutrition [19]. |
Synergistic Absorption and Cooperative Effects
Synergistic interactions occur when one mineral enhances the absorption or biological utilization of another, often through roles as enzymatic cofactors or by positively influencing shared metabolic pathways.
Copper and Iron: Copper acts as an essential cofactor for several enzymes critical to iron metabolism, including ceruloplasmin (Cp) and hephaestin. These multicopper ferroxidases are required for the efficient export of iron from cells (enterocytes and macrophages) into the circulation by oxidizing Fe²⁺ to Fe³⁺ for loading onto transferrin. Copper deficiency can therefore lead to iron accumulation in tissues and functional iron deficiency anemia, despite adequate iron stores [20].
Vitamin C and Iron: Ascorbic acid (Vitamin C) is a potent enhancer of non-heme iron absorption. It acts as a reducing agent, converting insoluble Fe³⁺ to the more soluble Fe²⁺ form, which is preferred by DMT1. Furthermore, vitamin C can form a chelate with iron that remains soluble in the alkaline environment of the duodenum, preventing its precipitation and increasing its pool for absorption [21].
Multiple Mineral Co-factors: The antioxidant enzyme superoxide dismutase (SOD) requires copper, zinc, and manganese as cofactors for its different isoforms (Cu/Zn-SOD and Mn-SOD), demonstrating a functional synergy in combating oxidative stress [22] [19]. Similarly, adequate zinc status is necessary for the synthesis of proteins involved in cellular iron storage and transport [20].
Table 2: Key Synergistic Mineral Interactions and Their Physiological Consequences
| Mineral Pair/Group | Site/Mechanism of Synergy | Physiological Impact |
|---|---|---|
| Copper & Iron | Cu is a cofactor for ferroxidases (ceruloplasmin, hephaestin) essential for Fe export [20]. | Improved Fe mobilization and circulation; Cu deficiency mimics Fe deficiency anemia [20]. |
| Vitamin C & Iron | Reduces Fe³⁺ to Fe²⁺ and forms absorbable iron-ascorbate chelate [21]. | Can significantly increase non-heme iron absorption, counteracting inhibitors like phytate [21]. |
| Zinc, Copper, Manganese | Act as cofactors for different isoforms of the antioxidant enzyme superoxide dismutase (SOD) [22] [19]. | Enhanced collective antioxidant defense and protection against oxidative cellular damage [19]. |
| Magnesium & Iron | Low Mg status can exacerbate Fe deficiency, though mechanisms are not fully elucidated [20]. | Adequate Mg may be necessary for optimal Fe metabolism and utilization. |
Quantitative Data on Mineral Bioaccessibility and Bioavailability
Understanding the inherent absorbability of minerals from food sources and the impact of processing is crucial for interpreting interaction studies. The following table summarizes quantitative data on mineral concentrations and absorption parameters.
Table 3: Mineral Compositions, Bioaccessibility, and Bioavailability Ranges in Leafy Vegetables (mg/100g dry weight, unless noted) [21]
| Mineral | Concentration Range in Leafy Vegetables | Bioaccessibility (%) | Bioavailability (%) | Impact of Processing |
|---|---|---|---|---|
| Iron (Fe) | 0.47 – 180.03 mg | 3.00 – 63.85% | 1.90 – 16.44% | Boiling loss: 24.16-71.54%. Steaming loss: 14.36-29.04%. Bioaccessibility can increase with processing. |
| Calcium (Ca) | 0.26 – 2455.00 mg | 3.00 – 75.80% | 0.70 – 40.00% | Boiling loss: 24.16-71.54%. Steaming loss: 14.36-29.04%. Bioaccessibility can increase with processing. |
| Zinc (Zn) | 0.06 – 56.10 mg | 2.00 – 69.00% | ~13.70% | Boiling loss: 24.16-71.54%. Steaming loss: 14.36-29.04%. Bioaccessibility can increase with processing. |
| Copper (Cu) | 0.01 – 16.00 mg | 5.70 – 75.50% | Not Specified | Boiling loss: 24.16-71.54%. Steaming loss: 14.36-29.04%. Bioaccessibility can increase with processing. |
| Selenium (Se) | 2.80 – 1100.60 μg | 10.80 – 90.00% | Not Specified | Boiling loss: 24.16-71.54%. Steaming loss: 14.36-29.04%. Bioaccessibility can increase with processing. |
Experimental Protocols for Assessing Mineral Interactions
In Vitro Bioaccessibility and Bioavailability Assays
Simulated gastrointestinal digestion models provide a rapid, cost-effective method for initial screening of mineral availability from food matrices and the impact of other dietary components.
Protocol: Static In Vitro Digestion Model [21] [23]
- Sample Preparation: Homogenize test food material. Precisely weigh a portion (typically 1-5 g) into a digestion vessel.
- Oral Phase: Add simulated saliva fluid (containing electrolytes and α-amylase) to the sample. Adjust pH to 7.0 and incubate at 37°C for 2-5 minutes with constant agitation.
- Gastric Phase: Add simulated gastric fluid (containing pepsin). Adjust pH to 2.0-3.0 with HCl. Incubate at 37°C for 1-2 hours with constant agitation.
- Intestinal Phase: Add simulated intestinal fluid (containing pancreatin and bile salts). Adjust pH to 7.0 with NaHCO₃. Incubate at 37°C for 2 hours with constant agitation.
- Centrifugation: Centrifuge the final digestate at high speed (e.g., 10,000 × g, 30 min, 4°C) to separate the soluble fraction (bioaccessible portion) from the solid residue.
- Analysis: The supernatant can be analyzed for bioaccessible mineral content using techniques like Atomic Absorption Spectroscopy (AAS) or Inductively Coupled Plasma Mass Spectrometry (ICP-MS). To estimate bioavailability, the supernatant can be further subjected to dialysis or Caco-2 cell uptake studies.
Assessing Mineral-Mineral Competition Using Caco-2 Cell Models
The human colon adenocarcinoma cell line (Caco-2), upon differentiation, exhibits an enterocyte-like phenotype and expresses key mineral transporters, including DMT1. It is a gold standard in vitro model for studying intestinal absorption and competitive interactions.
Protocol: Competitive Uptake Assay in Caco-2 Cells [21]
- Cell Culture: Grow Caco-2 cells in Transwell inserts until fully differentiated (typically 21 days), forming a tight monolayer.
- Treatment: Prepare treatment solutions in a physiologically relevant buffer (e.g., HBSS) containing:
- A fixed, tracer-level concentration of a radioisotope or stable isotope of the "target" mineral (e.g., ⁵⁹Fe or ⁵⁴Fe).
- Varying concentrations (0-10x molar excess) of the "competing" mineral (e.g., Zn, Ca, Cd).
- Uptake Experiment: Apply the treatment solution to the apical side of the Caco-2 monolayer. Incubate at 37°C for a defined period (e.g., 30-60 minutes).
- Termination and Analysis: Remove the apical solution and wash the monolayer thoroughly with ice-cold buffer to stop uptake and remove non-specifically bound minerals. Lyse the cells and analyze the isotope content in the lysate using a gamma counter (for radioisotopes) or ICP-MS (for stable isotopes).
- Data Interpretation: A significant decrease in the uptake of the target mineral with increasing concentrations of the competitor indicates competitive inhibition. Data can be analyzed using Michaelis-Menten kinetics to determine the nature of inhibition.
In Vivo Assessment and Serum Analysis
Human and animal studies are ultimately required to validate in vitro findings and understand systemic outcomes.
Protocol: Stable Isotope Studies for Mineral Absorption in Humans [24]
- Isotope Administration: Administer orally a test meal intrinsically or extrinsically labeled with a stable isotope of the mineral of interest (e.g., ⁵⁷Fe, ⁶⁷Zn). A different isotope of the same mineral can be administered intravenously to correct for pool kinetics (double isotope technique).
- Sample Collection: Collect blood, urine, and/or fecal samples at baseline and over a specified period (e.g., several days for blood and urine; complete fecal collection until isotope excretion is negligible).
- Sample Analysis: Isolate the mineral from the samples (e.g., iron from blood via chromatography, minerals from urine/fecal ash via acid digestion). Analyze isotopic enrichment using ICP-MS.
- Calculation: Fractional absorption of the oral isotope is calculated based on the appearance in the blood, the shift in isotopic ratio in urine, or the disappearance from the feces.
Protocol: Serum Trace Mineral Analysis for Status Assessment [19]
- Sample Collection: Collect venous blood serum samples using trace-element-free collection tubes to prevent contamination.
- Sample Preparation: Dilute serum samples with a dilute acid solution (e.g., nitric acid) or a specialized diluent. Alternatively, digest samples with high-purity nitric acid and hydrogen peroxide in a microwave-assisted digestion system.
- Analysis: Analyze the prepared samples using ICP-MS. This technique allows for the simultaneous, highly sensitive quantification of multiple essential (e.g., Zn, Cu, Se, Fe) and toxic (e.g., Pb, Cd, As, Hg) elements.
- Data Interpretation: Compare serum concentrations to established reference ranges to identify deficiencies, excesses, or imbalances. Correlate mineral levels with health outcomes or dietary intake data.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Reagents and Materials for Mineral Absorption and Interaction Research
| Research Reagent / Material | Function and Application in Research |
|---|---|
| Caco-2 Cell Line | A human colorectal adenocarcinoma cell line that, upon differentiation, forms a polarized monolayer with enterocyte-like properties. It is a standard in vitro model for studying intestinal mineral transport, permeability, and competitive interactions [21]. |
| Stable Isotopes (e.g., ⁵⁷Fe, ⁷⁰Zn, ⁶⁵Cu) | Non-radioactive isotopes used to intrinsically or extrinsically label foods. They allow for the precise tracking and quantification of mineral absorption, distribution, and retention in human and animal studies without radiation exposure [24]. |
| Inductively Coupled Plasma Mass Spectrometry (ICP-MS) | An analytical technique characterized by exceptionally low detection limits and the ability to measure multiple elements simultaneously. It is the gold standard for quantifying trace minerals and their stable isotopes in biological (serum, urine, tissues) and food samples [19]. |
| Simulated Gastrointestinal Fluids | Standardized solutions containing electrolytes and enzymes (e.g., pepsin, pancreatin, bile salts) that mimic the composition of human saliva, gastric, and intestinal juices. They are used in in vitro digestion models to assess mineral bioaccessibility [21] [23]. |
| Chelated Minerals (e.g., Amino Acid Chelates like Bisglycinates) | Mineral complexes where the metal ion is bound to a chelating agent like an amino acid. These are used in research to study formulations that resist inhibition by dietary factors (e.g., phytate) and demonstrate superior absorption and gastrointestinal tolerability compared to inorganic salts [25]. |
| Phytase Enzyme | An enzyme that hydrolyzes phytic acid (a potent mineral absorption inhibitor). Used in research (both in vitro and in vivo) to study the effect of phytic acid degradation on the bioaccessibility of minerals like iron and zinc, often in the context of cereal- and legume-based diets [23]. |
Visualization of a Mineral Interaction Research Workflow
The following diagram outlines a logical, multi-method workflow for investigating mineral-mineral interactions, from initial screening to clinical validation.
The efficacy of nutrients and bioactive compounds is not solely determined by the ingested dose but by their bioavailability—the proportion that is absorbed, transported to target tissues, and utilized in physiological processes [18]. This bioavailability is profoundly influenced by a constellation of host-specific factors, creating significant inter-individual variability in response to identical nutrient intakes. The central thesis of mineral absorption research is expanding to acknowledge that these host factors are not merely confounding variables but are primary determinants of nutritional outcome. This whitepaper provides an in-depth technical examination of three critical host-specific domains: genetic polymorphisms in genes governing absorption, distribution, metabolism, and excretion (ADME); the composition and function of the gut microbiota; and physiological changes across the human life cycle. A comprehensive understanding of these factors is paramount for researchers and drug development professionals aiming to develop targeted interventions, personalize nutritional strategies, and accurately predict compound efficacy.
Genetic Polymorphisms and Nutrient Bioavailability
Single nucleotide polymorphisms (SNPs) can alter the function of proteins involved in nutrient metabolism, thereby modifying an individual's nutritional requirements and response to supplementation.
Key Genetic Variants and Associated Nutrients
Table 1: Key Genetic Polymorphisms Influencing Nutrient Bioavailability
| Gene | Nutrient/Compound | Key Polymorphism(s) | Functional Consequence |
|---|---|---|---|
| VDR | Vitamin D | rs2228570 (FokI), rs1544410 (BsmI), rs11568820 (Cdx2) [26] [27] | Altered receptor function; impacts bone mineral density and response to Vitamin D supplementation [26] [28] [27]. |
| CYP2R1, CYP24A1, GC | Vitamin D | Multiple SNPs [26] | Affects synthesis, degradation, and transport of Vitamin D; influences baseline serum 25(OH)D and response to supplementation [26]. |
| FADS1 | Omega-3 Fatty Acids | rs174537 [28] | Modifies fatty acid metabolism; individuals with the G allele have increased OA risk with low omega-3 intake (OR: 1.45) [28]. |
| IL-6 | Antioxidants / General Inflammation | rs1800795 [28] | Carriers of the GG genotype with low antioxidant intake show elevated inflammatory markers and disease risk (OR: 1.60) [28]. |
| SULTs, UGTs, COMT | Dietary (Poly)phenols | 16 significant SNPs out of 88 studied [29] | Impacts phase II conjugation metabolism, altering plasma levels and urinary excretion of phenolic metabolites [29]. |
| MTHFR | Folate | Not specified in results | Associated with postmenopausal osteoporosis risk, implicating role in nutrient metabolism and bone health [27]. |
Experimental Protocols for Genotype-Phenotype Association
To investigate the impact of genetic polymorphisms on nutrient response, researchers employ carefully designed clinical trials and genetic association studies.
Protocol 1: Vitamin D Supplementation and Genotyping
- Population: Recruit healthy adults or elderly individuals, stratified by ethnicity. Exclude individuals with conditions or medications known to affect Vitamin D status [26].
- Intervention: Administer a standardized Vitamin D supplementation regimen (e.g., cholecalciferol). The dosage and duration should be pre-specified.
- Outcome Measurement: Collect blood samples pre- and post-intervention. Quantify serum 25-hydroxyvitamin D (25(OH)D) concentrations using a validated method (e.g., LC-MS/MS) [26].
- Genotyping: Isolate genomic DNA from participant blood or saliva. Perform genotyping for pre-selected SNPs in Vitamin D metabolism genes (e.g., VDR, CYP2R1, CYP24A1, GC) using TaqMan assays, microarrays, or sequencing.
- Statistical Analysis: Use linear regression models to test for associations between genotype and the change in serum 25(OH)D concentration, adjusting for covariates such as age, sex, and BMI [26].
Protocol 2: Cross-Sectional Gene-Nutrient Interaction Study
- Study Design: Recruit participants in a case-control or cross-sectional manner, such as 250 osteoarthritis patients and 250 healthy controls [28].
- Dietary Assessment: Administer validated food frequency questionnaires (FFQs) to estimate habitual intake of nutrients of interest (e.g., omega-3 fatty acids, vitamin D, antioxidants).
- Genotyping and Outcome: Genotype for target polymorphisms (e.g., FADS1 rs174537, VDR rs2228570). The primary outcome is disease risk or a relevant biomarker.
- Statistical Analysis: Employ multivariable logistic regression to test for gene-nutrient interactions on disease risk, calculating odds ratios (ORs) and confidence intervals (CIs) while adjusting for confounders [28].
Gut Microbiota as a Modulator of Host Nutrition
The gut microbiota functions as a metabolic organ, significantly influencing the bioavailability of dietary compounds through transformation, synthesis, and interaction with host absorption pathways.
Mechanisms of Microbial Influence
The gut microbiota modulates host nutrition via several key mechanisms:
- Fermentation and SCFA Production: Gut bacteria ferment dietary fibers resistant to human digestion, producing short-chain fatty acids (SCFAs) like acetate, propionate, and butyrate. These SCFAs serve as an energy source for colonocytes, regulate immune function, and influence host metabolism [30] [4].
- Metabolite Sensing: Host G-protein coupled receptors (GPCRs), such as GPCR41 and GPCR43, sense microbial metabolites including SCFAs. This sensing regulates the secretion of gut hormones from enteroendocrine cells, which in turn influences systemic metabolic homeostasis [4].
- Biotransformation of Complex Molecules: The microbiota metabolizes a wide range of compounds that human enzymes cannot process. For instance, many dietary (poly)phenols reach the colon intact and are transformed by gut bacteria into smaller, more bioavailable catabolites that are then absorbed and further conjugated by host phase II enzymes [29] [4].
Life Stage and Dysbiosis
The composition and function of the gut microbiota are not static but evolve throughout life, with aging being a critical period of change.
- Age-Related Shifts: In the elderly, gut microbiota often undergoes detrimental changes, including reduced diversity, an increase in pro-inflammatory taxa, and impaired production of beneficial metabolites like SCFAs [30].
- Consequences of Dysbiosis: These changes contribute to a state of chronic low-grade inflammation ("inflammaging"), immune senescence, and are associated with age-related conditions such as cognitive decline, metabolic disorders, and frailty [30]. Dysbiosis can also weaken intestinal barrier function, further impacting nutrient absorption and systemic health [30] [31].
Table 2: Gut Microbiota Shifts Across Life Stages and Dietary Influences
| Life Stage / Condition | Microbiota Characteristics | Impact on Bioavailability & Health |
|---|---|---|
| Early Infancy | Dominance of Bifidobacteria; highly malleable [30] [31]. | Critical for immune and metabolic maturation; establishes long-term health trajectories [31]. |
| Adulthood | High diversity and stability; dominated by Firmicutes and Bacteroidetes [30]. | Optimal metabolic function, SCFA production, and nutrient absorption [30]. |
| Elderly / Aging | Reduced diversity; loss of beneficial taxa; increased pro-inflammatory microbes [30]. | Contributes to inflammation, immune-senescence, and reduced absorption of key nutrients [30]. |
| Mediterranean / Plant-Based Diet | Enhances beneficial microbes (e.g., SCFA-producers); promotes microbial diversity [30]. | Improves metabolic health, reduces inflammation, and supports healthy aging [30]. |
| Western Diet | Promotes dysbiosis; reduces microbial diversity; increases inflammation-associated taxa [30]. | Accelerates aging processes and increases risk of age-related diseases [30]. |
Life Stage as a Determinant of Bioavailability
Physiological changes throughout the human life cycle directly impact the absorption and utilization of nutrients.
Critical Windows of Development and Aging
- The First 1000 Days: This period from pregnancy to age two is a critical window of developmental plasticity. Maternal health, diet, mode of delivery (vaginal vs. C-section), and infant feeding (breastfeeding vs. formula) collectively program the infant's gut microbiota, immune system, and metabolic pathways, with lasting effects on health and nutrient metabolism [31].
- Pregnancy and Lactation: This life stage is characterized by an increased absorptive capacity for many nutrients to meet the demands of the developing fetus and infant [18].
- Elderly: Older adults often exhibit a reduced ability to absorb certain vitamins and minerals. This can be exacerbated by age-related dysbiosis, bacterial overgrowth, and polypharmacy, where several medications are known to reduce vitamin absorption [30] [18].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents and Materials for Bioavailability Studies
| Reagent / Material | Function / Application | Example Use Case |
|---|---|---|
| TaqMan Assays / SNP Microarrays | Genotyping of specific single nucleotide polymorphisms (SNPs). | Determining participant genotype for genes like VDR or FADS1 in association studies [26] [28]. |
| Validated Food Frequency Questionnaires (FFQs) | Assessment of habitual dietary intake of nutrients and bioactive compounds. | Quantifying intake of omega-3 fatty acids, vitamin D, or antioxidants in cohort studies [28]. |
| LC-MS/MS (Liquid Chromatography-Tandem Mass Spectrometry) | Highly sensitive and specific quantification of nutrient metabolites in biological fluids. | Measuring serum 25(OH)D levels or (poly)phenol metabolites in plasma/urine [29] [26]. |
| 16S rRNA Gene Sequencing Reagents | Profiling the composition and relative abundance of bacterial taxa in gut microbiota. | Characterizing gut microbiome differences between age groups or dietary interventions [30] [32]. |
| Intestinal Organoids | In vitro model of the human intestinal epithelium for mechanistic studies. | Testing the effects of specific microbial metabolites on enterocyte proliferation and gene expression [4]. |
| GPCR-Specific Cell-Based Assays | Screening for metabolite-receptor interactions and signaling activation. | Identifying which microbial SCFAs activate GPCR41 or GPCR43 [4]. |
Visualizing Host-Microbiota-Nutrient Interactions
The following diagram synthesizes the core relationships and pathways through which host genetics, gut microbiota, and life stage converge to influence nutrient bioavailability and ultimate health outcomes.
Diagram Title: Host-Microbiota-Nutrient Interaction Network
This diagram illustrates the interconnected network determining nutrient bioavailability. Diet provides substrates for both the Host (via direct absorption, ADME) and the Gut Microbiota. The microbiota, in turn, metabolizes compounds and influences host Immune and Metabolic Maturation. Host Genetics directly modulates ADME processes, while Life Stage programs immune/metabolic maturation and shapes the microbiota composition. The interplay of these factors ultimately determines Health Outcomes and Disease Risk.
The Strong Ion Difference (SID) theory, pioneered by Peter Stewart, provides a physicochemical framework for understanding acid-base balance by emphasizing the deterministic role of electrolytes and weak acids in regulating hydrogen ion concentration. This paradigm shift from traditional bicarbonate-centered models offers a more mechanistic approach to analyzing acid-base disorders, with profound implications for understanding systemic mineral status. This whitepaper examines the core principles of SID theory, its clinical applications for assessing mineral balance, and its integration within broader research on mineral absorption inhibitors and enhancers. Designed for researchers, scientists, and drug development professionals, this technical guide synthesizes current evidence, presents quantitative data in structured tables, and provides detailed methodologies for experimental investigation of acid-base-mediated mineral regulation.
Traditional approaches to acid-base balance have primarily utilized the Henderson-Hasselbalch equation to categorize four primary acid-base disturbances: respiratory acidosis (increased PCO₂), respiratory alkalosis (decreased PCO₂), metabolic acidosis (decreased extracellular base excess), or metabolic alkalosis (increased extracellular base excess) [33]. While clinically useful when plasma protein concentrations are normal, this approach becomes limited when protein and phosphate concentrations are markedly abnormal, leading to potentially erroneous conclusions about the cause of acid-base disturbances [33].
The Strong Ion Difference (SID) theory, developed by Stewart, represents a fundamental shift in acid-base physiology by applying physicochemical principles to biological systems. This framework identifies three independent variables that determine pH in aqueous solutions: the carbon dioxide tension (pCO₂), the total concentration of non-volatile weak acids (Aₜₒₜ), and the strong ion difference (SID) – the difference between the sum of all strong cations (e.g., Na⁺, K⁺, Ca²⁺, Mg²⁺) and the sum of all strong anions (e.g., Cl⁻, lactate⁻, ketoacid anions) [33] [34]. According to this model, hydrogen ion concentration and bicarbonate levels are dependent variables that change in response to alterations in these three independent factors.
The relationship between SID and mineral metabolism is particularly relevant for nutritional science and drug development. Minerals exist predominantly as strong ions in biological fluids, and their absorption, distribution, and excretion directly influence systemic acid-base balance through effects on SID. Conversely, acid-base status can significantly modulate mineral bioavailability, tissue distribution, and physiological utilization, creating a complex regulatory network with important implications for health and disease.
Core Principles and Mathematical Framework
Independent Variables in Acid-Base Equilibrium
The Strong Ion Theory posits that blood pH is simultaneously regulated by three independent variables:
Strong Ion Difference (SID): Representing the net electrical effect of completely dissociated ions, SID constitutes the primary determinant of pH in physiological systems. The SID is calculated as:
SID = ([Na⁺] + [K⁺] + [Ca²⁺] + [Mg²⁺]) - ([Cl⁻] + [other strong anions])
In healthy individuals, SID typically maintains a value of approximately 40-44 mEq/L, which creates a slight electrical imbalance that favors alkalinity [34].
Total Concentration of Non-Volatile Weak Acids (Aₜₒₜ): This variable represents the aggregate concentration of partially dissociated acids, primarily albumin, phosphate, and sulfate. These substances function as important buffers, with their protonation state varying with pH. Alterations in plasma protein concentration significantly impact Aₜₒₜ and consequently affect acid-base balance.
Partial Pressure of Carbon Dioxide (pCO₂): As the only independent volatile acid-base variable, pCO₂ regulates the respiratory component of acid-base balance. The dissolved CO₂ interacts with water to form carbonic acid, which dissociates into bicarbonate and hydrogen ions according to the equilibrium:
CO₂ + H₂O ⇌ H₂CO₃ ⇌ H⁺ + HCO₃⁻ [35]
Quantitative Relationships in Strong Ion Theory
Table 1: Primary Acid-Base Disturbances in the Strong Ion Framework
| Disorder Type | Primary Defect | Independent Variable Change | pH Effect |
|---|---|---|---|
| Respiratory acidosis | Increased pCO₂ | pCO₂ ↑ | Decrease |
| Respiratory alkalosis | Decreased pCO₂ | pCO₂ ↓ | Increase |
| Strong ion acidosis | Decreased [SID] | [SID] ↓ | Decrease |
| Strong ion alkalosis | Increased [SID] | [SID] ↑ | Increase |
| Nonvolatile buffer ion acidosis | Increased [Aₜₒₜ] | [Aₜₒₜ] ↑ | Decrease |
| Nonvolatile buffer ion alkalosis | Decreased [Aₜₒₜ] | [Aₜₒₜ] ↓ | Increase |
| Temperature acidosis | Increased body temperature | Temperature ↑ | Decrease |
| Temperature alkalosis | Decreased body temperature | Temperature ↓ | Increase |
The mathematical foundation of SID theory derives from the principles of electroneutrality and conservation of mass in aqueous solutions. The fundamental equation representing this relationship is:
[SID] + [H⁺] - [HCO₃⁻] - [A⁻] - [CO₃²⁻] - [OH⁻] = 0
Where [A⁻] represents the dissociated form of weak acids. This equation must always hold true in biological fluids, meaning that any change in SID must be compensated by changes in the other components to maintain electrical neutrality.
The following diagram illustrates the deterministic relationships between independent and dependent variables in the Strong Ion Theory framework:
SID Theory in Mineral Absorption and Bioavailability Research
Mineral Interactions with Acid-Base Balance
Minerals constitute the primary strong ions that determine SID in biological systems. The systemic acid-base status directly influences mineral absorption, excretion, and metabolic utilization through multiple mechanisms:
Renal Mineral Handling: The kidney maintains acid-base homeostasis by modulating the excretion of minerals, particularly through the regulation of chloride reabsorption and sodium-potassium exchange. In metabolic acidosis, the renal tubules increase hydrogen ion secretion while simultaneously enhancing calcium and magnesium excretion, potentially leading to negative mineral balance [36] [1].
Gastrointestinal Absorption: The strong ion environment within the intestinal lumen significantly affects mineral solubility and transport. An acidic luminal pH generally enhances the absorption of cationic minerals (e.g., calcium, magnesium, iron, zinc) by promoting their solubility and facilitating passive paracellular transport [1]. This principle is exploited in mineral supplementation strategies that utilize acidic salts (e.g., calcium chloride, zinc sulfate) to improve bioavailability.
Systemic Mineral Redistribution: Acid-base disturbances can trigger the release of minerals from physiological buffers, particularly bone. Chronic metabolic acidosis promotes bone resorption through activation of osteoclast activity, leading to the release of carbonate, citrate, and calcium phosphate salts that serve as systemic buffers [36].
Inhibitors and Enhancers of Mineral Absorption
The SID framework provides a mechanistic understanding of how dietary factors influence mineral status. The most significant mineral absorption inhibitors include:
Phytic Acid (Myoinositol Hexakisphosphate): As the principal storage form of phosphorus in cereals, legumes, oil seeds, and nuts, phytic acid chelates cationic minerals (Fe, Zn, Ca, Mg, Mn) to form insoluble salts with poor bioavailability [9] [37]. Phytic acid content varies considerably across food sources, with concentrations ranging from 0.18-1.67 g/100g in millet to 2.56-8.7 g/100g in rice bran [9].
Table 2: Phytic Acid Content in Selected Food Groups
| Food Category | Specific Food | Phytic Acid Content (g/100g dry weight) | References |
|---|---|---|---|
| Cereals | Wheat bran | 2.1 - 7.3 | [9] |
| Rice bran | 2.56 - 8.7 | [9] | |
| Maize germ | 6.39 | [9] | |
| Legumes | Kidney beans | 0.61 - 2.38 | [9] |
| Soybeans | 1.0 - 2.22 | [9] | |
| Oilseeds | Sesame seed | 1.44 - 5.36 | [9] |
| Nuts | Almonds | 0.35 - 9.42 | [9] |
Dietary Electrolyte Balance: The dietary electrolyte balance (dEB), calculated as Na⁺ + K⁺ - Cl⁻, represents a practical application of SID principles in nutrition science. Diets with low dEB (high chloride relative to sodium and potassium) induce metabolic acidosis that impairs mineral retention, while high dEB diets promote mineral conservation [1]. This relationship is particularly important in animal nutrition, where manipulation of dEB is routinely used to optimize calcium status during lactation and egg production.
Experimental Methodologies and Analytical Approaches
Protocol for Assessing Acid-Base Status Using SID Theory
Materials and Equipment:
- Blood gas analyzer with ion-selective electrode capability
- Centrifuge for serum separation
- Spectrophotometer or autoanalyzer for protein and phosphate quantification
- Laboratory information system for data integration and SID calculation
Procedure:
Sample Collection and Preparation:
- Collect arterial or venous blood using heparinized syringes for blood gas analysis.
- Collect additional blood samples in serum separator tubes for electrolyte and protein analysis.
- Process samples within 15 minutes of collection to prevent glycolysis-induced changes.
- Centrifuge serum samples at 3000 × g for 10 minutes and aliquot for individual analyses.
Analytical Measurements:
- Analyze blood pH, pCO₂, and pO₂ using a calibrated blood gas analyzer [35].
- Measure serum sodium, potassium, chloride, calcium, and magnesium concentrations using ion-selective electrodes or spectrophotometric methods.
- Quantify serum albumin using bromocresol green method or immunoturbidimetry.
- Determine serum phosphate concentration using spectrophotometric molybdate-based assays.
SID Parameter Calculation:
- Calculate apparent SID (SIDₐ) as: SIDₐ = ([Na⁺] + [K⁺] + [Ca²⁺] + [Mg²⁺]) - ([Cl⁻] + [lactate⁻])
- Calculate effective SID (SIDₑ) using the formula: SIDₑ = [HCO₃⁻] + [albumin⁻] + [phosphate⁻] where [albumin⁻] = [albumin in g/L] × (0.123 × pH - 0.631) and [phosphate⁻] = [phosphate in mmol/L] × (0.309 × pH - 0.469)
- Determine the strong ion gap (SIG) as: SIG = SIDₐ - SIDₑ (SIG identifies the presence of unmeasured anions in plasma) [33] [34].
Protocol for Investigating Mineral Bioaccessibility
Materials:
- In vitro digestion system simulating gastric and intestinal phases
- pH-stat titration equipment
- Dialysis membranes with appropriate molecular weight cut-off
- Inductively coupled plasma mass spectrometry (ICP-MS) for mineral quantification
Procedure:
Sample Preparation:
- Grind food samples to particle size <0.5 mm to ensure homogeneity.
- Weigh 2.0 g samples in triplicate for digestion experiments.
Simulated Gastrointestinal Digestion:
- Gastric Phase: Add 20 mL simulated gastric fluid (0.15 M NaCl, pH 2.0) containing pepsin (3.2 g/L). Incubate at 37°C for 60 minutes with continuous agitation.
- Intestinal Phase: Adjust pH to 6.5-7.0 with NaHCO₃ solution. Add 20 mL simulated intestinal fluid containing pancreatin (1.9 g/L) and bile salts (12 g/L). Incubate at 37°C for 120 minutes with continuous agitation [38].
Mineral Bioaccessibility Assessment:
- Centrifuge digested samples at 10,000 × g for 30 minutes at 4°C.
- Collect supernatant (soluble fraction) for mineral analysis.
- Analyze mineral content in original samples and soluble fractions using ICP-MS.
- Calculate bioaccessibility as: Bioaccessibility (%) = (Mineral in soluble fraction / Total mineral in sample) × 100
The following workflow diagram illustrates the integrated experimental approach for assessing mineral status and bioaccessibility within the SID framework:
The Scientist's Toolkit: Essential Research Reagents and Methodologies
Table 3: Essential Research Reagents for SID and Mineral Absorption Studies
| Reagent/Method | Function | Application Notes |
|---|---|---|
| Ion-selective electrodes | Quantification of specific strong ions (Na⁺, K⁺, Cl⁻, Ca²⁺) | Essential for accurate SID calculation; requires regular calibration and quality control |
| Blood gas analyzer | Simultaneous measurement of pH, pCO₂, HCO₃⁻ | Critical for acid-base status assessment; sample integrity is paramount |
| ICP-MS (Inductively Coupled Plasma Mass Spectrometry) | Ultra-sensitive quantification of mineral elements | Enables simultaneous multi-mineral analysis at trace concentrations |
| Simulated gastrointestinal fluids | In vitro digestion model for bioaccessibility studies | Composition standardized to mimic human digestive conditions |
| Phytase enzymes | Hydrolyzes phytic acid to improve mineral bioavailability | Used to study dephytinization strategies in food processing |
| Dialysis membranes | Separation of soluble mineral fractions during digestion | Molecular weight cut-off determines which complexes are analyzed |
| Albumin and globulin standards | Protein quantification for Aₜₒₜ determination | Essential for accurate calculation of effective SID |
| SIG calculation software | Automated computation of strong ion gap | Reduces calculation errors in complex clinical or research datasets |
Research Implications and Future Directions
The integration of Strong Ion Theory with mineral absorption research presents significant opportunities for advancing nutritional science and therapeutic development:
Personalized Nutrition Strategies: Understanding individual variations in acid-base regulation and mineral metabolism can inform targeted nutritional interventions for populations with increased mineral requirements (e.g., osteoporosis, anemia, growth stages) [36] [37].
Drug-Mineral Interactions: Pharmaceutical development must consider how therapeutic agents affect acid-base balance and subsequent mineral status. Diuretics, antacids, and proton-pump inhibitors all influence SID through effects on renal mineral handling or gastrointestinal absorption.
Bioavailability Optimization: Food processing techniques (fermentation, germination, enzymatic treatment) that reduce phytate content can significantly improve mineral bioavailability [9] [38]. The SID framework provides a mechanistic basis for evaluating the efficacy of these processing strategies.
Advanced Analytical Approaches: Emerging technologies like thermal infrared hyperspectral (TIH) imaging show promise for rapid, non-destructive mineral analysis in complex biological matrices [39]. Such methodologies could revolutionize how mineral status is assessed in both research and clinical settings.
The Strong Ion Theory provides a robust physicochemical framework for understanding the intricate relationships between acid-base balance and mineral status. By focusing on the deterministic role of strong ions and weak acids, this approach offers mechanistic insights that extend beyond the descriptive capabilities of traditional bicarbonate-centered models. The integration of SID principles with research on mineral absorption inhibitors and enhancers enables a more comprehensive understanding of mineral metabolism, with significant implications for nutritional science, therapeutic development, and clinical practice. As analytical methodologies advance, the application of SID theory to mineral research will continue to generate valuable insights for addressing global challenges related to mineral deficiencies and metabolic disorders.
Advanced Assessment Techniques and Pharmaceutical Enhancement Strategies
Within mineral nutrition research, accurately determining bioavailability is paramount for understanding how effectively dietary minerals are absorbed, utilized, and retained by the body. Bioavailability encompasses digestibility, absorption, and metabolic utilization, and its measurement is critical for developing nutritional guidelines and therapeutic interventions. This guide details three core methodological paradigms—Balance Studies, Ileal Digestibility, and Stable Isotope Tracers—framed within the context of researching inhibitors and enhancers of mineral absorption. These methodologies enable scientists to quantify the complex journey of minerals from ingestion to incorporation into functional pools, providing insights that are foundational for advancements in clinical nutrition and drug development.
Core Measurement Paradigms
Balance Studies
The Nitrogen Balance method has been the historical gold standard for estimating protein requirements and, by extension, can inform studies on mineral co-factor relationships. The technique aims to measure the balance between nitrogen intake and nitrogen loss to determine the dietary intake required to maintain lean body mass [40]. While directly focused on nitrogen, the principles are applicable to mineral balance studies, where the intake and output of a specific mineral are measured.
- Principle: The body is in a state of equilibrium when the intake of a nutrient equals its loss. A positive balance indicates retention (anabolism), while a negative balance indicates loss (catabolism).
- Procedure: Subjects consume a controlled diet with a fixed amount of the mineral of interest. All excretory outputs—urine, feces, and sweat—are collected for a precise period. The total mineral content in the excreta is analyzed and subtracted from the intake.
- Limitations: The method requires meticulous control over diet and complete collection of excreta, which is challenging outside metabolic wards. It may overestimate retention if routes of loss (e.g., sweat, skin, menstrual blood) are not fully accounted for. Furthermore, balance studies do not provide information on true absorption, tissue-specific utilization, or internal metabolic recycling [40].
Ileal Digestibility
This paradigm provides a more precise measure of true digestibility by focusing on the site of absorption, the small intestine, thereby excluding the confounding effects of colonic fermentation and microbial activity.
- Principle: True digestibility is calculated by measuring the amount of a nutrient that disappears from the gastrointestinal tract before the end of the small intestine (ileum).
- Procedure: The gold-standard approach requires intubation of the terminal ileum in healthy subjects or the use of ileostomized patients. The test meal, often containing an non-absorbable marker (e.g., chromium oxide or polyethylene glycol), is administered. Ileal effluents are collected over a specific period, and the mineral content in the effluent is analyzed. The digestibility is calculated based on the difference between ingested and ileal-excreted mineral, corrected for the recovery of the marker [41].
- Limitations: The procedure is highly invasive, limiting its use in vulnerable populations. It also disrupts normal gastrointestinal physiology and requires a clinical setting, making it less suitable for large-scale or long-term studies.
Stable Isotope Tracers
Stable isotope methodologies represent a significant advancement, allowing for non-invasive, highly precise measurements of absorption, distribution, metabolism, and excretion of minerals in humans. These techniques are particularly powerful for studying the kinetics of mineral metabolism.
- Principle: A known quantity of a mineral enriched with a stable isotope (e.g., ⁵⁸Fe, ⁶⁷Zn, ⁴⁴Ca) is administered orally or intravenously. The dilution of this tracer in biological fluids (blood, urine) or its appearance in target tissues is monitored over time using mass spectrometry.
- Key Approaches:
- Dual-Stable Isotope Technique: This is a particularly powerful non-invasive method for measuring absorption. One stable isotope (e.g., ⁵⁷Fe) is administered orally, while a different isotope (e.g., ⁵⁸Fe) is administered intravenously. By comparing the enrichment of both isotopes in blood samples, the fractional absorption of the oral dose can be calculated without the need for fecal collection [41] [42].
- Arterial-Venous (A-V) Difference Method: This approach measures the net uptake or release of a nutrient across a specific tissue bed, such as muscle. Catheters are placed in an artery and a draining vein (e.g., femoral), and blood flow is measured. The A-V difference in mineral concentration, multiplied by blood flow, provides a direct measure of net tissue balance [40].
Table 1: Comparison of Bioavailability Measurement Paradigms
| Paradigm | Key Metric | Key Advantages | Key Limitations | Suitability for Mineral Research |
|---|---|---|---|---|
| Balance Studies | Whole-body retention/balance | Non-invasive; provides whole-body data; long-term studies possible [40]. | Incomplete collection leads to errors; does not measure true absorption [40]. | Low to Moderate: Useful for gross retention but lacks mechanistic insight. |
| Ileal Digestibility | True pre-cecal digestibility | Accurate measure of true absorption; excludes colonic interference [41]. | Highly invasive; not suitable for all populations; disrupts normal physiology [41]. | High for absorption studies, but practicality is low. |
| Stable Isotope Tracers | Fractional absorption, metabolic flux, tissue-specific uptake | Non-invasive; highly precise; provides kinetic data; can be used in vulnerable populations [41] [40] [42]. | High cost of isotopes and equipment (MS); complex data interpretation [40]. | Very High: Gold standard for human studies of mineral absorption and metabolism. |
Methodologies in Detail
Experimental Protocol: Dual-Stable Isotope Tracer Study
The following protocol, adapted from studies on protein digestibility, outlines a generalized procedure for a mineral absorption study using the dual-stable isotope method [41] [42].
1. Participant Preparation:
- Recruit participants based on the study criteria (e.g., age, health status). Obtain informed consent.
- Participants should fast overnight and refrain from strenuous exercise prior to the study day to standardize metabolic baseline.
- Perform baseline blood and urine collection.
2. Isotope Administration:
- Oral Dose: Administer a test meal containing the mineral of interest, intrinsically labeled with one stable isotope (e.g., ⁶⁷Zn). The meal should be consumed entirely.
- Intravenous Dose: Simultaneously or shortly after, administer a different isotope of the same mineral (e.g., ⁷⁰Zn) via a primed, constant intravenous infusion. The IV dose serves as a reference for calculating absorption and controlling for post-absorption metabolism and excretion.
3. Sample Collection:
- Collect serial blood samples (e.g., at 0, 30, 60, 120, 180, 240, 360, and 480 minutes post-dosing) from an arterialized venous line.
- Collect total urine and fecal outputs for a set period (e.g., 3-5 days) to monitor isotope excretion.
4. Sample Analysis:
- Process blood samples to isolate plasma or serum. Digest fecal and urine samples as necessary.
- Analyze the isotopic enrichment in the collected samples using Inductively Coupled Plasma Mass Spectrometry (ICP-MS).
5. Data Calculation:
- Fractional absorption of the oral mineral is calculated based on the ratio of the oral and intravenous tracer isotopes appearing in the blood or remaining in the body, correcting for isotopic excretion in urine and feces.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Reagents and Materials for Bioavailability Studies
| Item | Function/Application | Example in Mineral Research |
|---|---|---|
| Stable Isotope Tracers | To trace the metabolic fate of minerals without radioactivity. | ⁵⁷Fe, ⁶⁷Zn, ⁴⁴Ca for studying iron, zinc, and calcium absorption, respectively [41] [40]. |
| Non-Absorbable Markers | To correct for gut transit time and incomplete fecal collection in digestibility studies. | Polyethylene Glycol (PEG), Chromium Oxide. Mixed with test meals to mark the passage of digesta [41]. |
| Inductively Coupled Plasma Mass Spectrometry (ICP-MS) | To measure with high sensitivity and precision the concentration and isotopic enrichment of minerals in biological samples. | The core analytical instrument for quantifying stable isotope ratios in blood, urine, and tissue samples [41] [42]. |
| Amino Acid Mixtures | Used in protein-focused studies to modulate muscle protein synthesis; relevant for mineral co-factor studies in enzymes. | 2H-cell free amino acid mix used as a reference for 100% bioavailability in protein digestibility studies [41] [42]. |
| Bioavailability Enhancers | Compounds that improve the absorption of a co-administered nutrient/drug. | Piperine (from black pepper) inhibits drug-metabolizing enzymes and gut efflux pumps, enhancing bioavailability of various compounds [43]. |
| Chelating Agents / Absorption Enhancers | To alter membrane permeability and enhance paracellular absorption. | EDTA, Chitosan. Used in research to study mechanisms of absorption and potential applications for improving mineral bioavailability [44] [43]. |
Application in Inhibitor and Enhancer Research
The precise nature of stable isotope tracers makes them ideal for quantifying the effects of compounds that inhibit or enhance mineral absorption. For instance, the effect of phytic acid (a potent inhibitor of iron and zinc absorption) or ascorbic acid (an enhancer of non-heme iron absorption) can be directly and accurately measured in humans.
Experimental Design:
- Crossover Design: The same subject receives the test mineral dose with and without the inhibitor/enhancer in a randomized order, separated by a washout period.
- Dual Isotope Method: The fractional absorption of the mineral is measured using the dual-isotope technique under both conditions.
- Data Analysis: The percentage change in fractional absorption between the control and intervention phases quantifies the efficacy of the enhancer or inhibitor.
Table 3: Quantifying Effects of Inhibitors and Enhancers on Mineral Absorption
| Mineral | Modulating Compound | Mechanism of Action | Measured Effect on Absorption (Example) |
|---|---|---|---|
| Iron (Non-heme) | Phytic Acid (IP-6) | Forms insoluble complexes in the gut, preventing uptake [45]. | Reduction in fractional absorption by >50% can be precisely measured with ⁵⁷Fe/⁵⁸Fe tracers. |
| Iron (Non-heme) | Ascorbic Acid | Reduces Fe³⁺ to Fe²⁺ and forms a soluble chelate, enhancing uptake [45]. | Increase in fractional absorption by 2-3 fold, quantifiable via stable isotopes. |
| Various Minerals/Drugs | Piperine | Inhibits digestive enzymes, gut wall metabolism (glucuronidation), and drug efflux pumps like P-glycoprotein [43]. | Can increase bioavailability of co-administered compounds by 30% to 200%. |
| Various | Chitosan / Surfactants | Acts as an absorption enhancer by transiently opening tight junctions between enterocytes (paracellular transport) [44] [43]. | Increased plasma appearance curve of a mineral tracer, indicating enhanced absorption rate and extent. |
The study of mineral absorption is pivotal for addressing global micronutrient deficiencies, which are associated with impaired cognitive development, anemia, and compromised immune function [7]. Research in this field is increasingly reliant on advanced simulation methodologies to predict and enhance nutrient bioavailability. Bioavailability refers to the proportion of an ingested nutrient that is absorbed, transported, and utilized in physiological functions or storage [18]. This whitepaper details the integrated use of in vitro (simulated laboratory digestion) and in silico (computer simulation) models, framing them within a broader thesis on identifying and leveraging inhibitors and enhancers of mineral absorption. These models provide a cost-effective, high-throughput, and ethical alternative to traditional in vivo studies, enabling researchers to dissect the complex interplay between food matrix, processing, and the human gastrointestinal environment [46] [47]. Their application is transforming the development of functional foods and therapeutic formulations by offering unprecedented insights into digestive kinetics and bioavailability.
In Vitro Digestion Models: Experimental Protocols for Mineral Bioavailability
In vitro digestion models simulate the human gastrointestinal tract to estimate the bioaccessibility—the fraction of a nutrient released from the food matrix during digestion and available for absorption—and bioavailability of minerals [7] [18]. These models are highly versatile, allowing for the systematic evaluation of how food processing and dietary components act as enhancers or inhibitors of mineral absorption.
Detailed Protocol: Assessing Mineral Bioavailability in Processed Lentils
The following methodology, adapted from a study on iron-biofortified lentils, provides a robust framework for evaluating the impact of processing on mineral bioavailability [7].
Objective: To determine the impact of boiling and fermentation on the bioaccessibility and bioavailability of Fe, Zn, Cu, Ca, S, and Mg in iron-biofortified lentils using an in vitro digestion model coupled with the Caco-2 cell bioassay.
Materials:
- Test Material: Iron-biofortified lentil flour, processed into Raw Lentil Flour (RLF), Boiled Lentil Flour (BLF), and Fermented Lentil Flour (FLF).
- Digestive Enzymes: Pepsin (from porcine gastric mucosa, ≥250 U/mg), pancreatin (from porcine pancreas).
- Cell Culture: Caco-2 cells (human colorectal adenocarcinoma line, HTB-37 from American Type Culture Collection).
- Cell Culture Medium: Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 10% fetal bovine serum, 1% antibiotic-antimycotic solution, and 1% HEPES (1 mol/L).
- Assay Kits: Human ferritin ELISA kit for measuring iron bioavailability.
Procedure:
- Sample Preparation:
- Boiling: Lentils are boiled in deionized water (1:5 w/v) at 95°C for 30 minutes, then dried and milled.
- Fermentation: Lentil flour is mixed with water (1:2 w/v) and inoculated with Lactobacillus plantarum. Fermentation proceeds at 37°C for 48 hours, after which the material is inactivated and freeze-dried.
In Vitro Digestion Simulation:
- The digested sample is centrifuged. The supernatant, representing the bioaccessible fraction, is collected for mineral analysis and the Caco-2 cell assay.
Caco-2 Cell Bioassay for Iron Bioavailability:
- Caco-2 cells are cultured in DMEM and seeded at a density of 500,000 cells per well on 12-well collagen-coated plates.
- Cells are differentiated for 13-15 days, with the medium changed every 2 days.
- Prior to the experiment, the cell monolayer is washed with PBS.
- The bioaccessible fraction from the in vitro digestion is applied to the cells and incubated for 48 hours.
- After incubation, cells are harvested, and lysates are prepared.
- Ferritin Formation Analysis: Cell ferritin levels are quantified using a human ferritin-specific ELISA kit. Ferritin concentration is normalized to total cell protein content (expressed as ng ferritin/mg protein). This serves as a direct biomarker for cellular iron uptake and bioavailability [7].
Key Findings from the Lentil Study
The application of this protocol revealed significant insights into how processing alters mineral bioavailability, as summarized in the table below.
Table 1: Impact of Processing on Mineral Bioaccessibility and Bioavailability in Iron-Biofortified Lentils [7]
| Mineral | Metric | Raw Lentil Flour (RLF) | Boiled Lentil Flour (BLF) | Fermented Lentil Flour (FLF) |
|---|---|---|---|---|
| Fe | Bioaccessibility | Not Reported | Lower than FLF | 76.1% (Highest) |
| Bioavailability | Not Reported | Moderate | 69.4% (Highest) | |
| Zn | Bioaccessibility | Not Reported | 62.6% (Highest) | Lower than BLF |
| Bioavailability | Not Reported | 52.3% (Highest) | Lower than BLF | |
| Cu | Bioaccessibility | Not Reported | 99.0% (Highest) | Lower than BLF |
| Bioavailability | Not Reported | 81.2% (Highest) | Lower than BLF | |
| Ca | Bioaccessibility | Not Reported | 60.2% (Highest) | Lower than BLF |
| Bioavailability | Not Reported | Lower than FLF | 50.3% (Highest) | |
| Caco-2 Ferritin (ng/mg protein) | Baseline | Moderate | 14.7 (Highest) |
The study demonstrated that fermentation was the most effective processing method for enhancing iron and calcium bioavailability, as confirmed by the highest ferritin formation in Caco-2 cells. Correlation analysis further identified that Total Phenolic Content (TPC) and Total Dietary Fiber (TDF) were negatively correlated with mineral bioavailability, highlighting their role as absorption inhibitors [7].
In Silico Models: Computational Prediction of Digestive Processes
In silico models leverage computational power to simulate digestion and predict nutrient bioavailability. These tools range from models simulating fluid dynamics in the gut to bioinformatics algorithms predicting protein cleavage patterns [48] [46] [47].
Advanced Workflow: Integrating CFD and Machine Learning for Starch Digestion
A cutting-edge methodology combines 3D Computational Fluid Dynamics (CFD) with machine learning to create physiologically accurate 1D intestinal models, moving beyond oversimplified simulations [48].
Diagram: Workflow for Developing a Physiologically-Based In Silico Digestion Model
This workflow integrates critical physiological factors (KPFs) such as duodenal posture, gastric acidity, and peristaltic velocity that are often neglected in simpler models. The CFD simulations generate high-fidelity data on digestion kinetics, which is used to train a Recurrent Neural Network (RNN). The RNN acts as a surrogate model that captures the complex, time-dependent relationships between physiology and digestion, ultimately enhancing the predictive accuracy of the more computationally efficient 1D model [48]. This system can subsequently be integrated with blood glucose-insulin interaction models for a holistic view of post-absorptive metabolism.
In Silico Models for Protein Digestibility
For protein quality assessment, in silico tools like BIOPEP-UWM and PeptideCutter are used to simulate enzymatic cleavage based on protein sequence and protease specificity [46] [47]. These tools predict the release of peptides and amino acids, providing insights into protein digestibility. Furthermore, molecular docking studies can predict the affinity of resulting peptides for intestinal transporters, offering a glimpse into their absorption potential [47]. While promising, these models face challenges in incorporating the effects of protein folding, post-translational modifications, and dynamic gut conditions, which limits their current regulatory acceptance as standalone tools [46] [47].
A Framework for Predictive Bioavailability Equations
To translate experimental and computational data into actionable tools, a systematic framework for developing predictive bioavailability equations has been proposed [24]. This framework is essential for creating standardized algorithms that can estimate nutrient absorption without the need for testing every single food product.
Table 2: Four-Step Framework for Developing Predictive Bioavailability Equations [24]
| Step | Action | Description and Application |
|---|---|---|
| 1 | Identify Key Factors | Systematically list dietary and compound-specific factors (e.g., phytate, ascorbic acid, food matrix) that influence the absorption of the target nutrient. This forms the theoretical basis of the model. |
| 2 | Conduct Literature Review | Perform a comprehensive review of high-quality human studies to gather quantitative data on the impact of the identified factors on absorption. This populates the model with evidence-based parameters. |
| 3 | Construct Predictive Equation | Develop a mathematical equation, often using the relative bioavailability compared to a standard reference material. This allows for the prediction of absorption without host-specific factors. |
| 4 | Validate the Equation | Test the predictive performance of the equation against new experimental or clinical data. This step is critical for establishing reliability and enabling translation into applications like food labeling. |
This framework aims to generate equations that can be used to formulate products with enhanced nutritional value, improve food labeling, and conduct more accurate dietary intake assessments [24].
The Scientist's Toolkit: Essential Research Reagents and Models
This section catalogs critical reagents, cell models, and computational tools essential for conducting research in simulated digestion and mineral absorption.
Table 3: Key Research Reagent Solutions for Mineral Absorption Studies
| Category | Item | Function and Application in Research |
|---|---|---|
| Cell Models | Caco-2 Cell Line | A human intestinal epithelial cell model used to assess iron bioavailability via ferritin formation and to study transport pathways [7]. |
| Enzymes & Kits | Pepsin & Pancreatin | Digestive enzymes used in in vitro simulations of gastric and intestinal phases to break down the food matrix and release minerals [7]. |
| Ferritin ELISA Kit | Immunoassay kit for quantifying ferritin protein levels in Caco-2 cell lysates, serving as a biomarker for cellular iron uptake [7]. | |
| TDF Assay Kit | Used to quantify Total Dietary Fiber, an antinutritional factor that can inhibit mineral absorption [7]. | |
| Software & In Silico Tools | CFD Software (e.g., ANSYS) | Simulates fluid dynamics and mass transfer in the gastrointestinal tract, accounting for complex physiology [48]. |
| BIOPEP-UWM | An online database and tool for simulating the proteolytic cleavage of proteins by specific enzymes, predicting digestibility [46]. | |
| GastroPlus | A PBPK modeling platform that can simulate gastrointestinal digestion and absorption of compounds, adapted from pharmaceutical sciences [47]. | |
| Model Organisms | Gallus gallus (Chicken) | An in vivo model used to validate in vitro and in silico findings on mineral bioavailability and its effects on biomarkers like hemoglobin [7]. |
Data Integration and Visualization
The true power of modern nutrition research lies in the integration of data from in vitro and in silico sources. For instance, the quantitative bioavailability data from lentil processing (in vitro) can be used to train and validate machine learning models (in silico) that predict the outcomes of other processing methods on different crops.
Diagram: Mineral Absorption Research Pathway from Food to Bioavailability
This diagram illustrates the complementary research pathway. The direct in vitro to Caco-2 cell path provides ground-truthed biological data. The in silico prediction path, informed by processing data, can be used to rapidly screen new conditions, the results of which are subsequently validated in the biological assay. This iterative loop, guided by an understanding of inhibitors and enhancers, dramatically accelerates the research and development cycle for improving mineral nutrition.
The efficacy of orally administered therapeutics and the bioavailability of essential minerals are critically dependent on their successful permeation across biological barriers. Within the context of mineral absorption research, specific agents can act as powerful enhancers, mitigating the effects of known inhibitors and improving nutrient uptake. This whitepaper provides an in-depth technical examination of three major classes of permeation enhancement technologies: surfactants, chelating agents, and mucoadhesive polymers. We explore their fundamental mechanisms, detailed experimental methodologies, and quantitative performance data, framing this discussion within the broader research on modulating mineral absorption. The insights are intended to guide researchers and drug development professionals in selecting and applying these technologies to overcome pervasive challenges in drug delivery and nutrient bioavailability.
Surfactants
Mechanisms of Action
Surfactants are amphiphilic molecules that enhance permeation primarily by reducing interfacial tension and modifying wettability. Their hydrophobic tails and hydrophilic heads allow them to assemble at interfaces, such as those between oil and water or a solid and a liquid. In the context of mineral absorption, this action can disrupt the intestinal mucus layer and fluidize membrane components, potentially facilitating the paracellular and transcellular transport of mineral ions.
A key mechanism is the reduction of interfacial tension (IFT). For instance, in enhanced oil recovery, the surfactant BS-12 achieved ultralow IFT on the order of 10⁻³ mN·m⁻¹ in low-salinity systems [49]. While this specific data point is from another field, it illustrates the profound effect surfactants can have on interfacial properties. In biological terms, such a reduction in IFT at the intestinal lumen-enterocyte interface can lower the energy barrier for permeation.
Furthermore, surfactants significantly alter surface wettability. Research on bitumen liberation has shown that the zwitterionic surfactant erucamidopropyl betaine (EDAB) can reduce the water contact angle on bitumen-coated surfaces from 91° to 80°, demonstrating a shift towards a more hydrophilic, water-wet state [50]. This change in wettability can be correlated to improved dispersion and detachment from surfaces, which, in a physiological context, could translate to reduced adhesion of inhibitory substances and improved access to absorptive surfaces.
Quantitative Data and Efficacy
The efficacy of surfactants is quantitatively governed by parameters such as Critical Micelle Concentration (CMC), surface tension at CMC (γ_CMC), and adsorption efficiency (pC20). The following table summarizes experimental data for various surfactants.
Table 1: Surface Activity Parameters of Selected Surfactants
| Surfactant | Type | CMC (mol·dm⁻³) | γ_CMC (mN·m⁻¹) | pC20 | Key Finding | Source |
|---|---|---|---|---|---|---|
| Erucamidopropyl Betaine (EDAB) | Zwitterionic | ~10⁻¹ mmol/L | N/R | N/R | Reduced bitumen-silica adhesion force to 0 nN | [50] |
| BS-12 | Amphoteric | 6.26 × 10⁻⁴ | 30.15 | 4.54 | Achieved ultralow IFT (10⁻³ mN·m⁻¹) with heavy oil | [49] |
| Sodium Dodecyl Sulfate (SDS) | Anionic | N/R | N/R | N/R | Slightly enhanced negative surface charge of bitumen | [50] |
N/R: Not explicitly Reported in the sourced context
Atomic Force Microscopy (AFM) studies provide direct, nano-scale evidence of surfactant action. In the presence of EDAB, the adhesion force between bitumen and silica surfaces was reduced from 15 nN to 0 nN for hydrophilic silica and from 50 nN to 0 nN for hydrophobic silica [50]. This dramatic reduction in adhesion force is a direct metric of how effectively surfactants can suppress unwanted interactions, a mechanism that could be leveraged to prevent the adhesion of mineral-binding inhibitors in the gut.
Experimental Protocol: Interfacial Tension and Wettability Alteration
Objective: To evaluate the efficacy of a surfactant in reducing interfacial tension and altering the wettability of a solid surface.
Materials:
- Surfactant solution of interest (e.g., BS-12, EDAB)
- Saline solution (e.g., 1 mM KCl to simulate ionic strength)
- Model oil (e.g., Gudao heavy oil for IFT) or bitumen-coated surface (for wettability)
- Tensiometer (for surface tension)
- Spinning drop tensiometer (for ultra-low IFT)
- Goniometer (for contact angle measurements)
- Solid substrates (e.g., silica slides, aged rock cores)
Methodology:
- Surface Tension and CMC Determination:
- Prepare a series of surfactant solutions at varying concentrations.
- Measure the surface tension (γ) of each solution using a tensiometer.
- Plot γ against the logarithm of concentration (lg c). The concentration at which the slope breaks and surface tension plateaus is the CMC. γ_CMC is the corresponding surface tension.
- Calculate pC20 (-log C20) from the plot, where C20 is the concentration required to reduce surface tension by 20 mN·m⁻¹.
Interfacial Tension (IFT) Measurement:
- Using a spinning drop tensiometer, place a droplet of the model oil in a tube filled with the surfactant solution.
- Rotate the tube at high speed and analyze the droplet shape to calculate the IFT between the oil and surfactant solution.
Wettability Alteration Assessment:
- Age solid substrates (e.g., glass slides) by coating them with a model crude oil or mineral-adherent compound.
- Treat the aged substrates with the surfactant solution.
- Using a goniometer, place a water droplet on the treated and untreated solid surfaces and measure the contact angle. A decrease in angle indicates a shift towards water-wettability.
Analysis: FT-IR spectroscopy can be used post-treatment to confirm surfactant adsorption on the solid substrate by identifying new characteristic peaks (e.g., C–N and C=O bond stretches for BS-12) [49].
Mucoadhesive Polymers
Theories and Mechanisms of Mucoadhesion
Mucoadhesive polymers are bio-polymers designed to adhere to the mucosal surface layer for an extended period, thereby increasing retention time and improving the bioavailability of therapeutic agents or nutrients at absorption sites [51] [52]. The adhesion process is governed by several interrelated theories.
Table 2: Key Theories of Mucoadhesion
| Theory | Core Concept | Applicability/Considerations |
|---|---|---|
| Wetting Theory | Adhesion results from the ability of a liquid or polymer to spread and wet the mucosal surface, driven by interfacial forces and surface tension. | Primarily applicable to liquid or low-viscosity systems. Spreadability is inversely related to the contact angle. |
| Adsorption Theory | Adhesion is caused by the formation of primary (ionic, covalent) and secondary (hydrogen bonds, van der Waals, electrostatic) bonds at the interface. | Secondary bonds are more desirable for reversible mucoadhesion. |
| Diffusion Theory | Adhesion occurs due to the interpenetration and entanglement of polymer chains and mucin glycoproteins at the mucosal surface. | The depth of penetration, dependent on polymer chain flexibility and diffusion coefficient, determines bond strength. |
| Fracture Theory | Relates to the force required to separate two adhered surfaces, measuring the mechanical strength of the mucoadhesive bond. | Most applicable to single-component, rigid mucoadhesive materials. |
The mechanism of mucoadhesion is generally described as a two-stage process:
- Contact Stage: An intimate contact is established between the mucoadhesive polymer and the mucosal membrane.
- Consolidation Stage: The mucoadhesive material is activated by moisture, which plasticizes the system, allowing for the formation of secondary bonds (e.g., hydrogen bonds, van der Waals forces) with the mucosal layer [52].
Experimental Protocol: Evaluating Mucoadhesive Strength
Objective: To determine the mucoadhesive strength of a polymer using a tensile strength test.
Materials:
- Mucoadhesive polymer (e.g., chitosan, gelatin, thiolated polymers)
- Freshly excised mucosal tissue (e.g., porcine intestinal mucosa)
- Phosphate buffer saline (PBS, pH 6.8) to simulate intestinal conditions
- Texture Analyzer or Universal Testing Machine
- Double-sided adhesive tape or cyanoacrylate glue
Methodology:
- Sample Preparation: Prepare polymer discs or films using standard compression or casting techniques.
- Tissue Preparation: Mount a piece of mucosal tissue onto a support (e.g., a glass vial lid) using an adhesive, ensuring the mucosal side is facing upward. Keep the tissue hydrated with PBS.
- Assembly: Attach the polymer sample to the probe of the texture analyzer using an adhesive.
- Contact and Application of Force: Lower the probe at a defined speed until the polymer makes contact with the mucosal tissue. Apply a pre-defined force (e.g., 0.5 N) for a set contact time (e.g., 5-10 minutes) to allow for bond formation.
- Tensile Measurement: After the contact time, reverse the probe direction and withdraw it at a constant speed. The force required to detach the polymer from the mucosal tissue is recorded as the maximum detachment force (F_max).
- Data Analysis: The mucoadhesive strength is calculated as the maximum detachment force (F_max) divided by the surface area of contact (A). Additional parameters like the Work of Adhesion (area under the force-distance curve) can also be calculated.
Application in Drug and Nutrient Delivery
Mucoadhesive polymers are deployed in various dosage forms, including tablets, patches, films, and gels, for buccal, nasal, vaginal, and ocular delivery [51]. By increasing residency time at the absorption site, these systems can enhance drug permeability and bypass first-pass hepatic metabolism, which is crucial for both drugs and certain mineral complexes [51] [52]. Their ability to modulate dosage form localization offers a strategic tool for targeting specific regions of the GI tract for mineral absorption, such as the duodenum for iron or calcium.
Chelating Agents
Mechanisms in Mineral Absorption
Chelating agents enhance mineral absorption by forming stable, soluble complexes with metal ions, thereby preventing their precipitation or interaction with dietary inhibitors in the gastrointestinal tract. In mineral absorption research, this is pivotal for countering the effects of known inhibitors.
For example, mineral ions like calcium and iron can form insoluble complexes with anionic metabolites in the colon. Short-chain fatty acids (SCFAs), produced by microbial fermentation of fiber, acidify the colonic environment. This acidification prevents the formation of these insoluble mineral complexes, thereby improving the extent of mineral absorption [53]. This represents a natural, physiological chelation-like mechanism.
Protein-derived chelating peptides represent an advanced and highly effective class of synthetic chelators. These peptides, produced from hydrolyzed food proteins, form stable, soluble complexes with metal ions. Their key advantages over traditional mineral supplements include superior stability, safety, and very high bioavailability [54]. They enhance mineral absorption by protecting the mineral from dietary inhibitors (e.g., phytate), maintaining it in a soluble form, and potentially facilitating its transport via peptide transporters in the intestine.
Experimental Insights and Biological Activities
Beyond enhancing absorption, metal-chelating peptides have been found to provide additional biological benefits, such as antioxidant and immunomodulatory activities [54]. The conformational relationship between the peptide's structure and its chelating efficacy is a critical area of research, as the spatial arrangement of amino acids determines the stability and bioavailability of the mineral-peptide complex.
The following diagram illustrates the logical workflow for researching and applying permeation enhancers, connecting their mechanisms to experimental evaluation and practical applications.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Reagents and Materials for Permeation Enhancement Research
| Item | Function/Application | Example Usage in Research |
|---|---|---|
| Zwitterionic Surfactants (e.g., EDAB) | Modulate surface wettability and reduce adhesion forces; stable under high salinity/temperature. | Studying bitumen-mineral detachment [50]; model for disrupting mineral-inhibitor adhesion. |
| Amphoteric Surfactants (e.g., BS-12) | Achieve ultralow interfacial tension and alter rock surface wettability in enhanced oil recovery. | Core flooding experiments to study multiphase flow and detachment efficiency [49]. |
| Chitosan | A natural mucoadhesive polymer; positively charged, binds to anionic mucin via electrostatic forces. | Formulating nanoparticles or films for buccal, nasal, or GI drug delivery to enhance residency [55]. |
| Atomic Force Microscopy (AFM) with Colloidal Probe | Quantitatively measure nanoscale adhesion and interaction forces between surfaces in liquid. | Directly measuring the reduction in bitumen-silica adhesion force after surfactant application [50]. |
| Texture Analyzer | Quantify the mechanical strength of mucoadhesive bonds (tensile strength, work of adhesion). | Evaluating the mucoadhesive force of polymer films or tablets on excised mucosal tissue [52]. |
| Protein-Derived Chelating Peptides | Form stable, soluble complexes with metal ions to enhance mineral solubility and bioavailability. | Investigating as a superior alternative to traditional mineral supplements for preventing deficiencies [54]. |
| FT-IR Spectrometer | Characterize chemical bond formation and adsorption mechanisms at solid-liquid interfaces. | Confirming surfactant adsorption on aged rock cores by identifying new functional groups [49]. |
Surfactants, mucoadhesive polymers, and chelating agents offer distinct and powerful mechanisms to overcome the pervasive challenge of poor permeation in drug and mineral delivery. Surfactants operate by fundamentally altering interfacial properties, mucoadhesive polymers by increasing residency time at absorption sites, and chelating agents by maintaining essential minerals in a bioaccessible state. The quantitative data and rigorous experimental protocols outlined in this whitepaper provide a foundational toolkit for researchers. Integrating these technologies, particularly in the context of countering mineral absorption inhibitors, holds significant promise for developing next-generation therapeutic formulations and nutritional interventions with enhanced efficacy and bioavailability.
In the context of mineral absorption research, a significant challenge lies in overcoming the biological and dietary barriers that limit nutrient bioavailability. Inhibitors such as phytates, oxalates, and tannins can bind to essential minerals, forming insoluble complexes that prevent their absorption in the gastrointestinal tract [25] [7]. Conversely, research focuses on identifying and utilizing absorption enhancers, including certain organic acids, peptides, and advanced delivery systems that protect minerals and facilitate their uptake [56] [18]. Novel delivery systems represent a technological frontier designed to address these challenges directly. These systems—encompassing lipid-based formulations, nanocarriers, and encapsulation technologies—leverage principles of material science and nanotechnology to shield bioactive compounds, enhance their stability, and improve their targeted delivery. For mineral absorption, this means developing strategies that can protect mineral ions from dietary inhibitors, enhance their solubility, and promote their transport across the intestinal epithelium [57] [56]. This whitepaper provides an in-depth technical examination of these advanced delivery systems, focusing on their design principles, functional mechanisms, and application in mineral nutrition for researchers and drug development professionals.
Lipid-Based Nanocarriers: Architectures for Protection and Delivery
Lipid-based nanocarriers offer versatile platforms for encapsulating both hydrophilic and hydrophobic bioactives, including minerals and their chelates. Their biocompatibility and ability to modify release profiles and enhance permeability make them particularly valuable for improving mineral bioavailability.
2.1 Nanocochleates: Structured Lipid Cylinders for Sustained Release Nanocochleates (NCCs) are a distinctive class of lipid-based carriers formed through the interaction of negatively charged phospholipids (e.g., phosphatidylserine) with divalent cations like calcium (Ca²⁺) or magnesium (Mg²⁺) [57]. This interaction induces the rolling of lipid bilayers into stable, cylindrical, multi-lamellar structures resembling a spiral or a scroll. Their unique architecture confers several advantages for mineral delivery:
- High Physicochemical Stability: The tightly packed, dehydrated lipid layers minimize water penetration, protecting encapsulated minerals from degradation in the gastrointestinal environment [57].
- High Encapsulation Efficiency: NCCs efficiently encapsulate minerals, either within their lipid bilayers or by incorporating pre-formed mineral chelates [57].
- Sustained Release Properties: Diffusion-controlled release kinetics help maintain optimal mineral levels over extended periods [57].
Table 1: Composition and Characteristics of Select Lipid-Based Nanocarriers
| Nanocarrier Type | Key Structural Components | Typical Size Range | Encapsulation Efficiency (Reported Examples) | Primary Mineral Delivery Advantages |
|---|---|---|---|---|
| Nanocochleates (NCCs) | Phosphatidylserine, Ca²⁺/Mg²⁺ | 50 nm - 1 μm | High for hydrophobic actives [57] | Superior GI stability; sustained release; protects from dietary inhibitors [57] |
| Solid Lipid Nanoparticles (SLNs) | Solid lipid core (e.g., triglycerides), surfactant | 50 - 500 nm | Variable (e.g., ~50-90% for various actives) | Controlled release; increased stability vs. liposomes [58] [59] |
| Nanoemulsions | Oil phase, water phase, emulsifier | 20 - 200 nm | High for lipophilic compounds [60] | Enhanced solubility and dispersion of lipophilic mineral carriers [60] |
2.2 Liposomes and Other Vesicular Systems Liposomes are spherical vesicles comprising one or more concentric phospholipid bilayers enclosing an aqueous core. This structure allows for the simultaneous encapsulation of hydrophilic minerals (in the aqueous core) and hydrophobic compounds (within the lipid bilayers). While conventional liposomes are limited by physicochemical instability in the gastrointestinal tract, advanced variants like ethosomes and transfersomes have been engineered with enhanced flexibility for improved permeability [58].
Encapsulation Technologies for Mineral Fortification
Encapsulation involves coating a core material (e.g., a mineral) with a wall material to create micro- or nano-scale particles. This process physically isolates the mineral from the surrounding food or digestive environment, preventing undesirable interactions.
3.1 Spray Drying for Iron Fortification A prominent application involves encapsulating iron and ascorbic acid (an absorption promoter) within a matrix of Brewer's Spent Grain Arabinoxylans (AXs) via spray drying [56]. The detailed methodology is as follows:
- Objective: To develop an iron fortifier with enhanced stability and bioaccessibility for extruded corn products [56].
- Wall Material Preparation: Arabinoxylans (AXs) are extracted from Brewer's Spent Grain using alkaline extraction followed by ethanol precipitation [56].
- Feed Solution Formulation: Iron sulfate (FeSO₄) and ascorbic acid (AA) are dispersed in an AX solution. A typical formulation uses an iron-to-AX ratio of 1:20 and an AA-to-Fe molar ratio of 1.5:1. Maltodextrin may be added to achieve a total solid content of 10% (w/v) [56].
- Spray Drying Process: The feed dispersion is atomized in a spray dryer (e.g., Yamato ADL311S) under optimized parameters: an inlet air temperature of 180°C, an outlet air temperature of 96°C, a feed rate of 3.1 mL/min, and a drying air flow rate of 357 L/h [56].
- Outcome: The process yields powdered microcapsules with an encapsulation efficiency of approximately 53% for ascorbic acid. The iron within these capsules demonstrates a bioaccessibility of around 20% after in vitro gastrointestinal digestion [56].
3.2 Mineral Chelation: A Biomimetic Encapsulation Approach Chelation mimics natural processes where minerals are bound to organic molecules, as seen in chlorophyll (magnesium chelate) and hemoglobin (iron chelate) [25]. Albion Minerals' bisglycinate chelates are a prime example, where a mineral ion is bound to two glycine molecules, forming a stable, neutral-charge ring structure.
Table 2: Performance Comparison of Mineral Forms in the Presence of Inhibitors
| Mineral Form | Bioavailability Relative to Conventional Form | Key Findings from Clinical/In Vitro Studies |
|---|---|---|
| Calcium Bisglycinate | Up to 2x higher absorption [25] | Twice the absorption of calcium citrate and carbonate [25]. |
| Magnesium Bisglycinate | Up to 3x higher absorption [25] | Three times better absorption than magnesium oxide or citrate [25]. |
| Ferrous Bisglycinate | Up to 5x higher absorption [25] | In the presence of phytates, absorption was five times more effective than ferrous sulfate; reduces GI adverse events by 64% [25]. |
This chelated structure protects the mineral from interacting with dietary anti-nutrients like phytates and oxalates in the GI tract. The complex remains intact until absorbed in the small intestine via amino acid transport pathways, leading to significantly enhanced bioavailability and superior gastrointestinal tolerability compared to inorganic salts [25].
Experimental Protocols for Assessing Mineral Bioavailability
Robust in vitro methodologies are essential for screening and developing effective mineral delivery systems before proceeding to costly human trials.
4.1 Protocol: In Vitro Bioaccessibility and Bioavailability Assessment This protocol is adapted from studies on iron-biofortified lentils and encapsulated iron [56] [7].
- Principle: Simulated gastrointestinal digestion followed by measurement of mineral release (bioaccessibility) and uptake using a Caco-2 cell model (bioavailability).
- Materials & Reagents:
- Test Sample: Processed food material (e.g., fermented lentil flour) or encapsulated mineral powder.
- Enzymes: Pepsin (from porcine gastric mucosa), pancreatin (from porcine pancreas), bile salts.
- Simulated Fluids: Saliva, gastric, and intestinal electrolyte stock solutions.
- Cell Culture: Caco-2 cells (human colorectal adenocarcinoma cell line), Dulbecco's Modified Eagle Medium (DMEM), fetal bovine serum, antibiotic-antimycotic solution.
- Assay Kits: Human ferritin ELISA kit.
- Procedure:
- In Vitro Digestion:
- Oral Phase: Incubate sample with simulated saliva (pH 7.0) for 2-5 minutes.
- Gastric Phase: Adjust to pH 3.0, add pepsin solution, and incubate for 1-2 hours at 37°C with agitation.
- Intestinal Phase: Adjust to pH 7.0, add pancreatin and bile salts, and incubate for 2 hours at 37°C with agitation [7].
- Bioaccessibility Analysis: Centrifuge the digest (e.g., 10,000 × g, 30 min). The mineral content in the supernatant (soluble fraction) represents the bioaccessible portion. Calculate bioaccessibility as: (Mineral in supernatant / Total mineral in sample) × 100 [7].
- Caco-2 Cell Bioassay for Iron Bioavailability:
- Culture Caco-2 cells to form a differentiated monolayer.
- Apply the bioaccessible fraction (supernatant from step 2) to the cells and incubate for 24-48 hours.
- Harvest cells, lyse, and measure ferritin concentration (a marker of cellular iron storage) via ELISA.
- Express results as ng ferritin/mg cell protein [7]. An increase in ferritin formation indicates higher iron uptake and bioavailability.
- In Vitro Digestion:
4.2 Data Interpretation: Studies using this protocol have demonstrated that processing methods significantly impact mineral availability. For example, fermented lentil flour showed higher iron bioaccessibility (76.1%) and provoked greater ferritin formation in Caco-2 cells (14.7 ng/mg protein) compared to boiled or raw flour, highlighting the role of fermentation in reducing anti-nutritional factors [7].
Visualization of Experimental Workflows and System Mechanisms
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Mineral Delivery Studies
| Reagent/Material | Function/Application | Specific Example & Rationale |
|---|---|---|
| Anionic Phospholipids | Structural component for nanocochleates & liposomes [57]. | Phosphatidylserine: Interacts with Ca²⁺ to induce bilayer rolling into nanocochleate structures [57]. |
| Amino Acid Chelates | Bioavailable mineral sources for direct use or further encapsulation [25]. | Ferrous Bisglycinate: Proven resistance to phytate inhibition; WHO's "fortificant of choice" for targeted applications [25]. |
| Dietary Inhibitors | For in vitro challenge models to test delivery system efficacy [25] [7]. | Phytic Acid/Sodium Phytate: Standard solution to simulate inhibitory conditions of plant-based diets [25]. |
| Caco-2 Cell Line | In vitro model of human intestinal epithelium for absorption studies [7]. | ATCC HTB-37: Well-characterized model; ferritin formation is a validated marker for cellular iron uptake [7]. |
| Biopolymer Wall Materials | Form the encapsulating matrix for spray drying or coacervation [56]. | Brewer's Spent Grain Arabinoxylans: Sustainable by-product; contains glucuronic acid for potential cation interaction [56]. |
| Digestive Enzymes | Critical components for simulated in vitro gastrointestinal digestion [7]. | Pepsin & Pancreatin: Standard enzymes to mimic gastric and intestinal phases of human digestion [7]. |
Lipid-based formulations, nanocarriers, and encapsulation technologies provide sophisticated solutions to the persistent challenge of mineral malabsorption. By shielding minerals from dietary inhibitors, enhancing their stability, and facilitating targeted release, these systems directly counter the factors that limit bioavailability. The continued refinement of these delivery platforms, coupled with robust and predictive in vitro assessment models, holds significant promise for developing next-generation fortified foods and therapeutic mineral supplements. Future research should focus on optimizing scalable production, ensuring long-term stability, and conducting clinical trials to validate efficacy, thereby translating these advanced technological concepts into tangible public health benefits.
In the context of research on inhibitors and enhancers of mineral absorption, achieving precise control over intestinal permeability is a paramount objective. Physical enhancement methods—encompassing magnetic, acoustic, and mechanical approaches—offer sophisticated, non-invasive means to transiently modulate biological barriers for improved nutrient and therapeutic uptake. These technologies function primarily by applying controlled physical forces to cellular membranes and tight junctions, thereby facilitating the paracellular and transcellular transport of compounds that typically exhibit poor bioavailability. This whitepaper provides an in-depth technical analysis of the core principles, experimental protocols, and key research tools underpinning these advanced methodologies. The focus is placed on their application within mineral absorption research, where the ability to temporarily and reversibly enhance permeability can illuminate fundamental transport mechanisms and validate novel enhancement strategies.
Acoustic Permeability Modulation
Core Principles and Mechanisms
Acoustic technologies, particularly those utilizing surface acoustic waves (SAWs), have emerged as precise, contact-free tools for modulating cellular functions through mechanical stimulation [61]. These platforms generate programmable mechanical forces at appropriate length and frequency scales, enabling highly specific interactions with cells. The primary mechanism involves the application of acoustic radiation force directly onto cell membranes, which transiently alters membrane permeability by activating mechanosensitive ion channels [62]. This activation triggers downstream mechanotransduction pathways, including cytoskeletal remodeling and increased intracellular calcium ion flux, which can facilitate enhanced uptake of minerals and other compounds.
The technology demonstrates high biocompatibility due to the known transport properties of acoustic waves in aqueous media, making it particularly suitable for biomedical applications [61]. Unlike cavitation-based ultrasound methods that rely on bubble collapse, SAW-based systems like the ChemoTAP platform operate at high frequencies (e.g., 9.63 MHz) to exert nanoscale precision with minimal cellular damage [62]. This approach allows for highly localized, tunable mechanical stimulation without requiring microbubbles or contrast agents, reducing potential off-target effects and simplifying experimental design.
Quantitative Efficacy Data
Table 1: Quantitative Outcomes of Acoustic Permeability Modulation (ChemoTAP System)
| Parameter Measured | Experimental Result | Cell Line/Model | Key Mechanism |
|---|---|---|---|
| Intracellular Calcium Influx | 2.73-fold increase [62] | HeLa cells | Activation of mechanosensitive ion channels |
| Membrane Permeability | 1.73-fold enhancement [62] | HeLa cells | Acoustic radiation force on membrane |
| Chemotherapy Apoptosis | 1.78-fold increase with cisplatin [62] | HeLa tumor cells | Mitochondrial membrane depolarization, ROS generation |
| Acoustic Pressure | ~10⁷ Pa on LiNbO₃ substrate [62] | In vitro setup | Standing Surface Acoustic Waves (SAWs) |
| Acoustic Radiation Force | Order of 10⁻⁵ N [62] | HeLa cells | Dominant force over acoustic streaming drag force |
Experimental Protocol: ChemoTAP System
Objective: To transiently enhance cell membrane permeability and study subsequent drug/mineral uptake using tunable acoustofluidic permeabilization.
Materials & Equipment:
- Piezoelectric substrate (Lithium Niobate, LiNbO₃)
- Pair of focused interdigital transducers (fIDTs)
- Glass-bottom Petri dish
- RF signal generator
- Inverted fluorescence microscope with camera
- COMSOL Multiphysics software for simulation
- Cell culture reagents and fluorescent dyes (e.g., Fluo-4 AM for calcium flux)
Methodology:
- fIDT Fabrication: Fabricate a pair of fIDTs on the LiNbO₃ substrate using standard lithographic patterning. The design should enable generation of standing SAWs at a resonant frequency of 9.63 MHz.
- Cell Preparation: Culture adherent cells (e.g., HeLa, Caco-2) on separate glass-bottom Petri dishes until 70-80% confluency.
- System Assembly: Align the cell-cultured Petri dish above the fIDT-patterned substrate, ensuring the focal region of the transducers overlaps with the area of interest containing the cells.
- Acoustic Stimulation:
- Apply a continuous or pulsed RF signal (e.g., 250 ms pulses) to the fIDT pair to generate standing SAWs.
- The resulting acoustic radiation force propagates into the Petri dish, directly acting on the adherent cells.
- Permeability Assessment:
- Load cells with a fluorescent indicator (e.g., Fluo-4 AM for Ca²⁺) prior to stimulation.
- Record real-time fluorescence changes to quantify ion influx or molecular uptake.
- Perform post-stimulation viability assays (e.g., MTT, Annexin V) to ensure reversible permeabilization.
- Data Analysis: Quantify fluorescence intensity changes, calculate fold-increases in ion flux, and perform statistical analysis on uptake and viability data.
Key Considerations: The acoustic radiation force (Fr) dominates over acoustic streaming drag force (Fd) by approximately 6 orders of magnitude in this configuration, ensuring precise mechanical stimulation is the primary mechanism [62].
Magnetic and Mechanical Permeability Modulation
While acoustic methods show strong experimental validation, magnetic and mechanical approaches represent complementary strategies for permeability enhancement.
Magnetic Permeability Modulation
Magnetic stress methods typically involve the addition of magnetic nanoparticles or microparticles to exert controlled mechanical forces on cellular structures [62]. When exposed to an external magnetic field, these particles can generate localized stress that disrupts tight junctions or induces transient pores in cell membranes. However, these techniques face significant challenges related to off-target accumulation, potential particle contamination, and difficulties in particle elimination from biological systems [62]. The requirement for introducing exogenous particles complicates experimental standardization and raises concerns for translational applications, making them less favorable compared to contact-free acoustic methods.
Mechanical Permeability Enhancement
Mechanical enhancement strategies encompass various approaches including microfluidic-based forces, hydrodynamic stress, and wall shear stress [62]. These techniques leverage fluid dynamics to apply controlled mechanical forces on cellular membranes, leading to transient disruption and enhanced permeability.
Table 2: Comparison of Physical Permeability Enhancement Methods
| Method | Key Mechanism | Advantages | Limitations/Challenges |
|---|---|---|---|
| Acoustic (SAW) | Acoustic radiation force activating mechanosensitive channels [62] | High spatial precision, contactless, tunable, no additives required | Requires specialized transducer fabrication |
| Magnetic Stress | Force application via magnetic particles under external fields [62] | Remote activation, potential for deep tissue targeting | Particle contamination risk, off-target accumulation, difficult elimination |
| Microfluidic Shear | Hydrodynamic forces disrupting membranes [62] | Can be integrated with lab-on-a-chip systems | Effectiveness varies by cell type, potential for uncontrolled damage |
| Ultrasonic Cavitation | Inertial cavitation from collapsing microbubbles [62] | Established ultrasound technology | Unstable, difficult to control, highly bubble-dependent |
Signaling Pathways in Acoustic Mechanotransduction
The application of acoustic forces triggers specific intracellular signaling cascades that mediate the permeability response. The following diagram illustrates the key pathway activated by acoustic stimulation in the ChemoTAP system.
Diagram 1: Acoustic Mechanotransduction Pathway for Permeability Modulation. This pathway illustrates how Surface Acoustic Wave stimulation leads to enhanced cellular effects through calcium-mediated signaling.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Physical Permeability Studies
| Item/Category | Specification/Example | Primary Function in Research |
|---|---|---|
| Piezoelectric Substrates | Lithium Niobate (LiNbO₃) [62] | Generating Surface Acoustic Waves (SAWs) via the inverse piezoelectric effect. |
| Interdigital Transducers | Focused IDTs (fIDTs) [62] | Converting electrical signals into mechanical SAWs; focusing acoustic energy. |
| Fluorescent Indicators | Fluo-4 AM [62] | Quantifying real-time changes in intracellular calcium ion concentration as a marker for permeability. |
| Permeation Enhancers | Sodium Caprate (C10) [63] | Chemical enhancer used for comparative studies; improves peptide stability and absorption. |
| Therapeutic Agents | Cisplatin, GLP-1 Analogues [62] [63] | Model compounds for validating enhanced permeability and uptake efficacy. |
| Cell Culture Models | HeLa, Caco-2 cell lines [62] | In vitro models for studying permeability mechanisms and screening enhancers. |
Integrated Experimental Workflow
A comprehensive research strategy for evaluating physical enhancement methods typically follows a systematic workflow from platform preparation to data analysis, as detailed below.
Diagram 2: Integrated Workflow for Physical Permeability Research. This workflow outlines the key stages in conducting research on physical permeability modulation, from device fabrication to final validation.
Physical enhancement methods represent a frontier technology for precise modulation of biological permeability. Acoustic approaches, particularly SAW-based platforms like ChemoTAP, currently offer the most sophisticated and controllable means for transient permeability enhancement with demonstrated efficacy in increasing molecular uptake. The integration of these physical methods with traditional chemical enhancers and formulation strategies presents a promising avenue for future research, particularly in the challenging field of mineral absorption where bioavailability limitations often hinder therapeutic and nutritional efficacy. As these technologies mature, their application within mineral absorption research will likely provide unprecedented insights into transport mechanisms and enable the development of next-generation enhancement strategies.
Strategies to Overcome Absorption Barriers and Enhance Mineral Bioavailability
Mineral deficiencies represent a significant global health challenge, often resulting not from insufficient dietary intake but from poor absorption and bioavailability due to the presence of antinutritional factors (ANFs) in plant-based foods [7]. The efficacy of mineral absorption is governed by a complex interplay of inhibitors and enhancers within the food matrix and the gastrointestinal tract. This technical guide examines three strategic intervention categories—food processing, fermentation, and synergistic nutrient pairing—that can be employed to optimize mineral bioavailability within the context of modern food science and nutritional therapeutics. These interventions target specific biochemical pathways and food matrix components that otherwise limit mineral solubility, transport, and systemic utilization [64] [65]. Understanding these mechanisms is crucial for researchers and drug development professionals seeking to develop targeted nutritional solutions for populations affected by micronutrient deficiencies.
Impact of Food Processing on Mineral Bioavailability
Mechanisms of Processing-Induced Enhancement
Conventional and emerging food processing techniques significantly influence mineral bioavailability primarily through the degradation of antinutritional factors and the disruption of the food matrix. Thermal processing methods, including boiling, steaming, and pressure cooking, denature proteins and dissociate mineral complexes, thereby increasing mineral solubility. Hydrothermal treatments have been demonstrated to reduce tannin levels by up to 46.09% and phytic acid by 31.94% in proso millet, substantially decreasing their mineral-chelating capacity [66]. The application of heat and moisture also gelatinizes starch granules, encapsulating minerals and potentially creating protective structures that enhance mineral stability during digestion. However, the specific effects vary considerably based on the processing parameters (temperature, duration, pressure) and the food matrix characteristics [66].
Comparative Analysis of Processing Techniques
Table 1: Impact of different processing methods on antinutritional factors and mineral bioaccessibility
| Processing Method | Reduction in Phytic Acid | Reduction in Tannins | Impact on Mineral Bioaccessibility | Key Findings |
|---|---|---|---|---|
| Boiling | 31-47% | 40-52% | Variable increase (Fe: 15-25%, Zn: 10-20%) | Effective for lentils and legumes; maintains mineral content [7] |
| Pressure Cooking | 35-55% | 45-60% | Moderate increase (Ca: 20-30%, Fe: 25-35%) | High efficiency in ANF reduction; may cause mineral leaching [66] |
| Steaming | 25-40% | 30-45% | Mild to moderate increase | Preserves water-soluble minerals better than boiling [66] |
| Roasting | 20-35% | 35-50% | Mild increase (Zn: 5-15%, Fe: 10-20%) | Dry heat may create insoluble mineral complexes in some matrices [66] |
| Blanching | 15-25% | 20-30% | Minimal to mild increase | Limited effectiveness; primarily used as a pretreatment [66] |
The selection of processing parameters must be optimized for specific food matrices, as excessive heat treatment can promote the formation of insoluble mineral complexes or catalyze reactions that reduce mineral bioavailability. For instance, prolonged high-temperature processing may bind minerals to dietary fiber or promote oxidation states with lower absorption efficiency (e.g., conversion of Fe²⁺ to Fe³⁺) [7] [66].
Fermentation as a Bioprocessing Strategy
Biochemical Mechanisms of Fermentation
Fermentation enhances mineral bioavailability through multiple synchronized biochemical pathways mediated by microorganisms. Lactic acid bacteria (LAB), yeasts, and filamentous fungi secrete a spectrum of enzymes including phytases, polyphenol oxidases, and cellulases that systematically degrade antinutritional factors [67]. Phytase enzymes catalyze the stepwise hydrolysis of phytic acid (myo-inositol hexakisphosphate), releasing bound minerals and decreasing the phytate:mineral molar ratios that are critical predictors of mineral absorption [64]. Simultaneously, microbial metabolism generates organic acids (lactic, acetic, propionic) that acidify the food matrix, further promoting mineral solubilization and creating an optimal environment for mineral absorption in the gastrointestinal tract [7] [67].
The fermentation process also modifies the structural components of the food matrix through microbial degradation of cell wall polysaccharides, thereby increasing porosity and facilitating mineral release during digestion. Specific strains of Lactobacillus plantarum, Lactobacillus rhamnosus, and Bacillus subtilis demonstrate particularly high phytase activity and organic acid production, making them optimal candidates for targeted fermentation protocols [68] [67].
Experimental Evidence and Efficacy
Table 2: Mineral bioavailability enhancement through fermentation of various food matrices
| Food Matrix | Fermentation Method | Bioaccessibility/Bioavailability Outcomes | Key Mechanisms |
|---|---|---|---|
| Iron-biofortified lentils | Natural fermentation (48-72h) | Fe bioaccessibility: 76.1% (vs 45-55% in raw); Ferritin formation in Caco-2 cells: 14.7 ng/mg protein [7] | Phytate degradation, organic acid production, phenolic transformation |
| Pearl millet (koko, zoomkoom) | Traditional spontaneous fermentation (24-48h) | Phytic acid reduction: 56.7-76.8%; Iron bioaccessibility: up to 21.8%; Zinc bioaccessibility: up to 42.2% [64] | Phytate hydrolysis, tannin reduction, fiber modification |
| Bean dregs & soybean meal | Solid-state fermentation (11 days, 34°C) | Mineral complexation rates: Fe (97.2%), Zn (95.9%), Ca (90.6%), Mn (94.6%), Cu (73.3%) [68] | Small peptide and organic acid production, mineral chelation |
| Vegetables (broccoli, carrot, beet) | Lactic acid fermentation (21 days) | Varied mineral retention; enhanced vitamin A and carotene in carrots and peppers [69] | Organic acid production, biocomponent transformation |
Recent research demonstrates that solid-state fermentation (SSF) generally outperforms submerged fermentation (SmF) for mineral bioavailability enhancement in cereal and legume matrices due to superior enzymatic activity and phenolic compound retention [67]. The complexation of minerals with microbial metabolites such as small peptides and organic acids during fermentation creates stable complexes that resist precipitation in the alkaline environment of the small intestine, thereby maintaining minerals in absorbable forms throughout the critical absorption window [68].
Synergistic Nutrient Pairing Strategies
Fundamental Synergistic Mechanisms
Synergistic nutrient pairing leverages biochemical interactions between specific nutrients to enhance absorption, cellular uptake, and physiological utilization. These partnerships function through several established mechanisms: (1) alteration of mineral oxidation states to improve solubility (e.g., vitamin C-mediated reduction of Fe³⁺ to Fe²⁺), (2) regulation of transport proteins and channels (e.g., vitamin D-induced expression of calcium-binding proteins), (3) provision of molecular ionophores to facilitate cellular mineral uptake (e.g., quercetin-enhanced zinc transport), and (4) protection against oxidation during digestion and transport (e.g., vitamin E preservation of omega-3 fatty acids) [70] [71].
The efficacy of these synergistic relationships is influenced by molar ratios, timing of co-consumption, and the overall composition of the meal matrix. For instance, the enhancing effect of vitamin C on non-heme iron absorption demonstrates a dose-response relationship, with 500 mg of ascorbic acid increasing iron absorption up to six-fold while moderate doses of 50-100 mg still provide significant enhancement [71].
Evidence-Based Synergistic Pairings
Table 3: Documented synergistic nutrient pairs and their mechanisms of action
| Nutrient Pair | Mechanism of Synergy | Absorption Enhancement | Food-Based Examples |
|---|---|---|---|
| Vitamin C + Non-heme Iron | Reduces Fe³⁺ to more soluble Fe²⁺; forms absorbable iron-ascorbate chelates | Up to 6-fold increase with 500mg vitamin C [71] | Spinach with lemon juice; lentils with tomatoes; fortified cereals with berries |
| Vitamin D + Calcium | Regulates expression of calcium transport proteins (calbindin) in intestinal mucosa | Significantly improved calcium absorption efficiency [70] [71] | Fatty fish with dairy; fortified plant milks with vitamin D |
| Turmeric + Black Pepper | Piperine inhibits glucuronidation in intestinal mucosa and liver, enhancing curcumin bioavailability | Up to 2000% increase in curcumin bioavailability [70] | Traditional spice combinations in curries |
| Omega-3 Fatty Acids + Vitamin E | Vitamin E prevents peroxidation of long-chain polyunsaturated fatty acids | Improved incorporation of EPA/DHA into cell membranes [71] | Salmon with spinach; walnuts with berries |
| Zinc + Quercetin | Quercetin acts as zinc ionophore, facilitating cellular zinc uptake | Enhanced intracellular zinc concentrations and immune support [71] | Apples with pumpkin seeds; onions with oysters |
The strategic combination of these synergistic pairs within meals or supplement formulations requires careful consideration of potential antagonistic interactions. For example, high-dose calcium supplementation can inhibit iron absorption when consumed simultaneously, suggesting that temporal separation of these minerals may be necessary for optimal bioavailability [71]. Similarly, tannin-rich beverages like tea and coffee can significantly decrease non-heme iron absorption when consumed with meals, necessitating at least a one-hour separation for iron-sensitive individuals [7] [71].
Experimental Methodologies and Research Tools
In Vitro Bioaccessibility and Bioavailability Assessment
The standardized protocol for assessing mineral bioaccessibility and bioavailability employs simulated gastrointestinal digestion coupled with Caco-2 cell models, providing a robust, reproducible, and ethically favorable alternative to human trials [7] [65].
Protocol for In Vitro Digestion and Bioaccessibility Determination:
- Oral Phase: Homogenize 5g test sample with 6mL simulated salivary fluid (pH 6.8-7.0) containing α-amylase (75 U/mL). Incubate 5 minutes at 37°C with continuous agitation.
- Gastric Phase: Adjust pH to 2.5-3.0 with HCl, add pepsin solution (2000 U/mL in final mixture). Incubate 2 hours at 37°C with continuous agitation.
- Intestinal Phase: Adjust pH to 6.5-7.0 with NaHCO₃, add pancreatin (100 U/mL trypsin activity) and bile salts (10mM final concentration). Incubate 2 hours at 37°C with continuous agitation.
- Bioaccessible Fraction Separation: Centrifuge intestinal digest at 10,000×g for 60 minutes at 4°C. Collect supernatant (bioaccessible fraction) for mineral analysis via ICP-OES or ICP-MS [7] [64].
Caco-2 Cell Bioassay for Iron Bioavailability:
- Cell Culture: Maintain Caco-2 cells in DMEM with 10% fetal bovine serum, 1% non-essential amino acids, and 1% antibiotic-antimycotic solution at 37°C in 5% CO₂.
- Differentiation: Seed cells at 50,000 cells/cm² on collagen-coated transwell inserts and culture for 14-21 days to achieve complete differentiation.
- Bioavailability Assay: Apply bioaccessible fraction (diluted in uptake buffer) to apical chamber. Incubate 24 hours at 37°C.
- Ferritin Analysis: Harvest cells, lyse, and measure ferritin concentration using ELISA. Normalize to total cellular protein (Bradford assay). Ferritin formation correlates with cellular iron uptake and serves as the primary bioavailability endpoint [7].
This integrated approach allows for high-throughput screening of multiple processing interventions and their effects on mineral bioavailability, with strong correlation to human absorption studies when properly validated [7] [65].
Research Reagent Solutions
Table 4: Essential research reagents and materials for mineral bioavailability studies
| Reagent/Material | Specifications | Research Application | Key Function |
|---|---|---|---|
| Caco-2 cell line | HTB-37, American Type Culture Collection | Intestinal absorption model | Differentiates into enterocyte-like cells; expresses brush border enzymes and transporters [7] |
| Simulated digestive enzymes | Pepsin (porcine gastric mucosa, ≥250 U/mg); Pancreatin (porcine pancreas, 4×USP) | In vitro digestion models | Mimics proteolytic and lipolytic activities of human GI tract [7] |
| ICP-OES/MS | Agilent 5110 ICP-OES or equivalent | Mineral quantification | Precise multi-element analysis at trace concentrations [66] |
| Phytic acid assay kit | Megazyme K-PHYT or equivalent | Antinutrient quantification | Enzymatic measurement of phytic acid in food matrices [64] |
| Ferritin ELISA kit | Human ferritin ELISA kit | Iron bioavailability assessment | Quantifies ferritin protein in Caco-2 cell lysates [7] |
| FTIR spectrometer | Nicolet iS50 or equivalent | Molecular complexation analysis | Identifies mineral-ligand binding through characteristic absorption bands [68] |
Visualization of Mechanisms and Workflows
Mineral Bioavailability Enhancement Pathways
Diagram 1: Integrated pathways for enhancing mineral bioavailability through dietary interventions
Experimental Workflow for Bioavailability Assessment
Diagram 2: Experimental workflow for assessing mineral bioaccessibility and bioavailability
The strategic integration of food processing, fermentation, and synergistic nutrient pairing offers a multifaceted approach to combating mineral deficiencies through enhanced bioavailability. The evidence presented demonstrates that these interventions operate through distinct yet complementary mechanisms to maximize mineral absorption from plant-based foods. Future research should focus on optimizing these interventions for specific population needs, developing standardized predictive models for mineral bioavailability [65], and exploring novel microbial strains with enhanced phytase and fermentation capabilities. Additionally, the application of emerging technologies such as pulsed electric field processing and ultrasound-assisted fermentation shows promise for further enhancing the efficacy of these dietary interventions while maintaining nutritional quality and sensory attributes [67]. As research in this field advances, the development of personalized nutritional approaches based on genetic polymorphisms in mineral transporters and metabolic pathways will represent the next frontier in addressing global mineral deficiencies.
The integration of probiotic and microbiome engineering represents a transformative approach for enhancing mineral bioavailability in the human gastrointestinal tract. This technical guide explores the mechanistic role of microbial phytases—enzymes produced by engineered probiotics that hydrolyze antinutritional phytic acid (phytate). By breaking down the stable phytate-mineral complexes that hinder mineral absorption, these enzymes significantly increase the bioavailability of essential minerals such as iron, zinc, calcium, and magnesium. Framed within a broader thesis on inhibitors and enhancers of mineral absorption, this review provides an in-depth analysis of phytase-producing microorganisms, genetic engineering strategies for enhancing phytase expression in probiotic chassis, detailed experimental protocols for assessing phytase activity and mineral solubilization, and the subsequent impact on human mineral homeostasis. The development of targeted probiotic interventions offers a promising strategy to mitigate widespread mineral deficiencies and represents a significant advancement in the field of nutritional science and therapeutic microbiology.
A significant challenge in human nutrition is the ubiquitous presence of phytic acid (myo-inositol hexakisphosphate, IP6) in plant-based diets. As the primary storage form of phosphorus in seeds, grains, and legumes, phytate accounts for 65% of phosphorus fertilizer deposition in soils and is a common component of human diets [72]. Its strong chelating capacity, derived from twelve replaceable protons, allows it to form stable, insoluble complexes with di- and trivalent cations including Zn²⁺, Fe²⁺/³⁺, Ca²⁺, Mg²⁺, Mn²⁺, and Cu²⁺ in the gastrointestinal tract [73]. These phytate-mineral complexes are largely unavailable for absorption in the human small intestine, leading to reduced bioavailability of essential micronutrients even when dietary intake appears sufficient [73] [74].
The consequences of this mineral sequestration are profound, contributing to the global burden of micronutrient malnutrition that affects approximately 2 billion people worldwide [3]. This is particularly problematic in developing countries where diets rely heavily on cereals and legumes, and for specific populations including vegetarians, vegans, women of childbearing age, and the elderly [74] [3]. While conventional strategies like food fortification and mineral supplementation have been implemented, their efficacy is often limited by the persistent presence of phytate [73].
Phytase Classification and Catalytic Mechanism
Phytases (myo-inositol hexakisphosphate phosphohydrolases) are a class of phosphatases that catalyze the step-wise hydrolysis of phytic acid, releasing inorganic phosphate and lower myo-inositol phosphates (IP5-IP1) [72] [75]. This dephosphorylation process dismantles the mineral-chelating capacity of phytate, thereby liberating bound minerals and making them available for absorption in the small intestine [72].
The enzymatic reaction occurs as follows: Phytate (IP6) + H₂O → myo-inositol pentakisphosphate (IP5) + Pᵢ → ... → myo-inositol + Pᵢ
The physicochemical properties of phytases, including their pH optimum, thermostability, and protease resistance, determine their efficacy in the gastrointestinal environment. Their activity is further influenced by dietary composition and gut conditions [72].
While phytases are present in plants, animals, and microorganisms, microbial phytases are particularly advantageous for biotechnological applications due to their catalytic efficiency, stability, and ease of production [72] [75]. The table below summarizes the principal microbial sources of phytases:
Table 1: Major Microbial Sources of Phytases and Their Characteristics
| Microbial Group | Representative Species | Key Characteristics | Applications |
|---|---|---|---|
| Fungi | Aspergillus oryzae, A. niger | High enzyme yield, acid tolerance, well-characterized genetics | Poultry feed, food processing [72] [75] |
| Bacteria | Bacillus subtilis, B. coagulans | Thermostability, protease resistance | Probiotic engineering, animal feed [3] |
| Lactic Acid Bacteria | Lactobacillus acidophilus, L. fermentum, L. plantarum | Generally Recognized As Safe (GRAS) status, human gut adaptation | Engineered probiotics, functional foods [3] |
Compared to plant-based phytases, microbial phytases demonstrate superior effectiveness in mineral solubilization [72]. Their production can be scaled using cost-effective fermentation processes with readily available substrates, making them economically viable for widespread application [72] [75].
Engineering Probiotics for Phytase Production and Mineral Solubilization
Probiotic Chassis Selection
The selection of an appropriate microbial chassis is critical for developing effective phytase-delivery probiotics. Ideal candidates possess GRAS status, a well-characterized genetic system, and the ability to survive and function in the human gastrointestinal environment [76] [77].
Table 2: Promising Probiotic Chassis for Engineering
| Probiotic Strain | Advantages | Engineering Examples | Relevant Applications |
|---|---|---|---|
| Lactococcus lactis | Food-grade probiotic; established secretion systems; FDA-authorized for therapeutic delivery [76] | Secretion of guided antimicrobial peptides (gAMPs) targeting H. pylori [76] | Precision antimicrobials; potential phytase delivery |
| Escherichia coli Nissle 1917 | Well-characterized genetics; robust gut colonization; engineered for diagnostic and therapeutic functions [77] | "i-ROBOT" platform for diagnosing, recording, and ameliorating inflammatory bowel disease [77] | Modular platform for biomarker-responsive metabolite delivery |
| Lactobacillus spp. | Native gut inhabitants; some strains produce phytase naturally; multiple genetic tools available [3] | Natural phytase production observed in L. acidophilus, L. fermentum, L. plantarum [3] | Potential for enhanced native phytase expression or heterologous phytase introduction |
Genetic Engineering Strategies
Advanced synthetic biology tools enable the precise engineering of probiotics for high-level phytase production and controlled activity in the gut.
- Heterologous Gene Expression: Cloning and expression of phytase genes from highly efficient producers (e.g., Bacillus or Aspergillus species) into probiotic chassis. This often involves codon optimization for the host and the use of strong, constitutive, or inducible promoters [76] [75].
- Secretory Systems: Utilizing native secretion signals (e.g., the usp45 signal peptide in L. lactis) to direct the synthesized phytase enzyme to the extracellular environment, enabling it to act on dietary phytate within the gut lumen [76].
- Regulatory Circuits: Implementing biomarker-responsive genetic circuits that control phytase expression. For instance, promoters induced by low pH (such as the P1 promoter used in L. lactis [76]) or specific microbial metabolites (e.g., thiosulfate-responsive systems as in E. coli Nissle [77]) can ensure context-specific enzyme production, conserving cellular energy and enhancing safety.
The following diagram illustrates a generalized genetic engineering workflow for creating a phytase-producing probiotic:
Experimental Protocols for Assessing Phytase Activity and Mineral Bioavailability
Protocol: Quantitative Phytase Activity Assay
Objective: To determine the enzymatic activity of phytase produced by engineered probiotics.
Materials:
- Sodium Phytate Solution: (5 mM) in appropriate buffer (e.g., 100 mM sodium acetate for pH 5.5, simulating gastric conditions) [72].
- Stopping Reagent: Trichloroacetic acid (TCA, 15% w/v).
- Colorimetric Reagent: Ammonium molybdate/ammonium vanadate solution for inorganic phosphate (Pᵢ) detection [72] [75].
- Recombinant Probiotic Culture: Supernatant (for secreted phytase) or cell lysate (for intracellular phytase).
Method:
- Reaction Setup: Mix 500 µL of sodium phytate solution with 500 µL of enzyme preparation (supernatant/lysate) in a microcentrifuge tube.
- Incubation: Incubate the reaction mixture at 37°C for 30 minutes.
- Reaction Termination: Stop the reaction by adding 1 mL of 15% TCA.
- Precipitation: Centrifuge at 10,000 × g for 5 minutes to precipitate denatured proteins.
- Phosphate Quantification: Mix 500 µL of clear supernatant with 1 mL of colorimetric reagent. Incubate for 10 minutes at room temperature.
- Absorbance Measurement: Measure the absorbance at 415 nm using a spectrophotometer.
- Calculation: Determine the Pᵢ concentration from a standard curve prepared with KH₂PO₄. One unit (U) of phytase activity is defined as the amount of enzyme that liberates 1 µmol of inorganic Pᵢ per minute under the assay conditions [72] [75].
Protocol: In Vitro Mineral Solubilization Assay
Objective: To evaluate the ability of engineered probiotics to solubilize minerals from a phytate-mineral complex.
Materials:
- Phytate-Mineral Complex: Precipitated by mixing solutions of sodium phytate and mineral salts (e.g., FeSO₄, ZnCl₂) at neutral pH [73].
- Simulated Gastric Fluid: (pH ~2-3) and Simulated Intestinal Fluid: (pH ~6.8-7.2).
- Engineered Probiotic: Live culture or cell-free supernatant.
- Inductively Coupled Plasma Optical Emission Spectrometry (ICP-OES) for mineral quantification.
Method:
- Digestion Simulation: Suspend the phytate-mineral complex in simulated gastric fluid, add the probiotic preparation, and incubate with shaking (37°C, 60 min).
- Absorption Simulation: Adjust the pH to intestinal conditions and continue incubation (37°C, 120 min).
- Separation: Centrifuge the mixture at high speed to separate the solubilized fraction (supernatant) from the insoluble complex (pellet).
- Analysis: Analyze the supernatant using ICP-OES to quantify the concentration of solubilized minerals (Fe, Zn, Ca, Mg) [73].
- Data Expression: Report results as the percentage of total mineral solubilized compared to a negative control (non-phytase producing strain).
Research Reagent Solutions
Table 3: Essential Research Reagents for Probiotic Phytase Studies
| Reagent / Material | Function / Application | Example / Specification |
|---|---|---|
| Sodium Phytate (IP6) | Standard substrate for phytase activity assays; preparation of phytate-mineral complexes. | Purity >95%; 5 mM solution in appropriate buffer [72]. |
| Expression Vector with Secretion Signal | Genetic construct for heterologous phytase expression and secretion in probiotic chassis. | pT1NX vector for L. lactis (uses usp45 secretion signal, P1 promoter) [76]. |
| Simulated Gastrointestinal Fluids | In vitro simulation of stomach and intestinal environments for activity and stability testing. | Gastric fluid (pH 2-3, pepsin); Intestinal fluid (pH 6.8-7.2, pancreatin) [73]. |
| Caco-2 Cell Line | Human epithelial colorectal adenocarcinoma cell model for studying mineral transport and absorption. | For in vitro validation of enhanced mineral uptake [73] [65]. |
Impact on Mineral Absorption and Homeostasis
The ultimate goal of probiotic-driven phytase production is to enhance mineral bioavailability. The mechanism by which this occurs involves a multi-step process, visualized below:
The efficacy of mineral absorption is critically dependent on the Phytate-to-Mineral Molar Ratio. Reducing this ratio below a critical threshold is key to improving bioavailability, as demonstrated by the following quantitative relationships:
Table 4: Impact of Phytate-to-Mineral Molar Ratio on Bioavailability
| Mineral | Critical PA:Mineral Molar Ratio | Effect on Bioavailability | Reference |
|---|---|---|---|
| Zinc (Zn) | > 20 | Zn absorption reduced to 22-34% | [73] |
| < 10 | Zn absorption increases to 20-40% | [73] | |
| Iron (Fe) | 1 : 1 | Fe uptake reduced by ~56% | [73] |
| 10 : 1 | Fe uptake reduced by ~73% | [73] | |
| General | High Ratio | Strongly inhibits absorption of Fe, Zn, Ca, Mg | [73] [74] [78] |
Probiotics enhance this process not only via phytase production but also through other mechanisms including the production of short-chain fatty acids (SCFAs) that lower gut pH, further favoring mineral solubility, and by generally improving gut health, which supports the function of enterocytes and mineral transport systems [3].
The engineering of probiotics to produce microbial phytases represents a sophisticated and targeted strategy to overcome the persistent challenge of mineral malabsorption caused by dietary phytate. This approach aligns perfectly with the broader research objective of identifying and leveraging potent enhancers of mineral absorption while mitigating the effect of inhibitors.
Future research should focus on several key areas:
- Strain Optimization: Isolating or engineering phytases with optimal activity and stability across the variable pH and enzymatic conditions of the human GI tract.
- Advanced Delivery Systems: Developing biomarker-responsive, self-regulating probiotic systems that produce phytase in a demand-dependent manner [77].
- Human Trials: Conducting rigorous, well-controlled human clinical trials to validate the efficacy of engineered phytase probiotics in improving mineral status across diverse populations.
- Cocktail Formulations: Exploring the use of probiotic cocktails that deliver multiple enzymes (e.g., phytases and cellulases) or combined therapies (e.g., probiotics with prebiotics like certain fibers that SCFAs can ferment) for a synergistic effect on mineral nutrition.
The integration of synthetic biology with nutritional science holds immense promise for developing next-generation probiotic solutions to address global micronutrient deficiencies, ultimately contributing to improved public health outcomes.
In the study of mineral absorption, the precise chemical form of a mineral is a critical determinant of its bioavailability. Mineral chelates and organic complexes represent advanced forms where essential metal ions are bound to organic ligands, a process designed to enhance stability and navigate the complex environment of the gastrointestinal tract. The Association of American Feed Control Officials (AAFCO) provides generic definitions for these compounds, yet the terminology can be vague. A fundamental distinction lies between a complex—a general term for a species formed when a metal ion reacts with a molecule or ion (a ligand)—and a chelate, which is a specific type of complex where the ligand bonds to the metal ion via two or more donor atoms, forming a stable, heterocyclic ring structure [79].
This ring structure, often five-membered for greatest stability, is the cornerstone of the chelate's function [79]. The ligands involved are typically classified by their donor atoms (e.g., oxygen, nitrogen, sulfur) and can be monodentate (one donor atom) or polydentate (multiple donor atoms, such as bidentate or hexadentate) [79]. Amino acids, which are bidentate ligands that bond via the oxygen of the carboxylic acid group and the nitrogen of the amino group, are ideal for this purpose. In contrast, a strong chelator like ethylenediaminetetraacetic acid (EDTA) is hexadentate but can form complexes so stable that mineral bioavailability is negligible [79]. True chelation is predominantly feasible with transition elements like copper, iron, manganese, and zinc, which possess the necessary physico-chemical characteristics to form coordinate covalent bonds [79].
Mechanisms of Enhanced Stability and Bioavailability
The enhanced bioavailability of chelated minerals is not attributable to a single mechanism but rather to a series of strategic advantages conferred by the chelate structure throughout the absorption pathway.
Protection from Dietary Antagonists
A primary mechanism is the physical protection of the mineral ion within the organic ligand shell. In the digestive tract, minerals are vulnerable to interactions with dietary components that can render them insoluble and unabsorbable. Phytic acid, a common compound in plant-based foods like wheat, is a potent mineral antagonist that complexes with minerals, preventing their intestinal assimilation [23]. The chelate's stable ring structure shields the mineral from phytic acid and other antagonists like oxalates, tannins, and fiber, thereby maintaining the mineral in a soluble and absorbable form [80] [81]. This protection minimizes the formation of insoluble precipitates, a common fate for inorganic mineral salts such as sulfates, oxides, and chlorides [80].
Facilitated Absorption Pathways
Once protected, chelated minerals leverage alternative absorption routes. Due to their structural similarity to peptides, certain amino acid chelates are recognized by peptide transport systems in the intestinal lining and are absorbed more efficiently than inorganic ions [81]. This pathway can be particularly advantageous as it may reduce competition for absorption with other minerals that use ion-channel transporters [80]. The overall result is a significant increase in bioavailability—defined as the fraction of an ingested nutrient that is absorbed, transported to target tissues, and utilized in metabolic processes or storage [18]. Research in animal nutrition consistently demonstrates that chelated minerals can exhibit 30-60% higher bioavailability compared to their inorganic counterparts [80].
Table 1: Key Characteristics of Mineral Forms
| Characteristic | Inorganic Minerals (e.g., Sulfates, Oxides) | Chelated Minerals (e.g., Amino Acid Chelates) |
|---|---|---|
| Chemical Structure | Simple ionic salts | Stable, heterocyclic ring structure with organic ligands |
| Stability in GI Tract | Low; highly reactive and prone to precipitation | High; protected from antagonists by ligand shell |
| Primary Absorption Pathway | Ion channels | Peptide transporters and ion channels |
| Bioavailability | Lower; highly variable | Higher; consistently 30-60% more bioavailable in studies |
| Interaction with Antagonists | High; readily binds to phytates and oxalates | Low; shielded from interactions |
| Gastrointestinal Irritation | More common | Reduced |
Experimental Evidence and Quantitative Outcomes
Robust experimental models, particularly in livestock, have quantified the benefits of chelated minerals, moving beyond simple growth metrics to biochemical and environmental outcomes.
A 2025 study on growing-finishing pigs provides a compelling comparative analysis. The research replaced 100% inorganic trace minerals (ITMs) with low-dose amino acid-chelated trace minerals (OTMs) and assessed a comprehensive set of parameters [82]. The results demonstrated that a low-dose OTM supplementation (15 mg/kg Fe, 4 mg/kg Cu, 12.5 mg/kg Zn, 5 mg/kg Mn) could sustain performance while significantly enhancing antioxidant status and trace mineral deposition in tissues, comparable to a high-dose ITM regime (75 mg/kg Fe, 10 mg/kg Cu, 65 mg/kg Zn, 25 mg/kg Mn) [82].
Table 2: Quantitative Outcomes from Pig Study: Low-Dose Organic vs. High-Dose Inorganic Minerals [82]
| Parameter | High-Dose Inorganic Minerals (HITM) | Low-Dose Organic Chelates (LOTM) | Statistical Significance |
|---|---|---|---|
| Growth Performance | Baseline | No adverse effect | Not Significant (p>0.05) |
| Liver CuZn-SOD Activity | Baseline | Significantly higher | p < 0.05 |
| Liver MDA Content | Baseline | Significantly lower | p < 0.05 |
| Muscle Fe Content | Baseline | Significantly higher | p < 0.05 |
| Fecal Cu Excretion | Baseline | Significantly reduced | p < 0.05 |
| Fecal Zn Excretion | Baseline | Significantly reduced | p < 0.05 |
| Fecal Mn Excretion | Baseline | Significantly reduced | p < 0.05 |
The data confirms that chelation enhances the bioefficacy of minerals, allowing for a substantial reduction in dietary inclusion levels—up to 80% for Zn and Mn—without compromising physiological function [82]. This has a direct and profound impact on environmental pollution, dramatically reducing the fecal excretion of these heavy metals [82] [80].
Methodologies for Assessing Chelate Stability and Bioavailability
Evaluating the efficacy of mineral chelates requires a multi-faceted experimental approach, from in vitro simulations to in vivo trials.
Protocol for In Vitro Bioaccessibility Assessment
This protocol simulates gastrointestinal conditions to estimate the fraction of a mineral released from its matrix for potential absorption [23].
- Step 1: Sample Preparation. The test material (feed or food) is ground to a standardized particle size to ensure uniform digestion.
- Step 2: Gastric Phase Simulation. The sample is incubated in a solution containing pepsin and adjusted to pH 2.0 with HCl, mimicking the stomach environment. This phase typically lasts 1-2 hours at 37°C with constant agitation.
- Step 3: Intestinal Phase Simulation. The pH is raised to 6.5-7.0 using a sodium bicarbonate buffer, and pancreatin and bile salts are added to simulate the duodenum. Incubation continues for another 2-4 hours.
- Step 4: Analysis. The digestate is centrifuged to separate the bioaccessible fraction (supernatant) from the sediment. The mineral content in the supernatant is quantified using techniques like Inductively Coupled Plasma Optical Emission Spectrometry (ICP-OES).
Protocol for In Vivo Balance Study
This is a classic method for measuring apparent absorption in living organisms and is considered highly informative [18] [82].
- Step 1: Animal Trial Design. Animals are allocated into treatment groups (e.g., inorganic vs. chelated mineral diets) in a controlled environment. The study should be approved by an animal ethics committee.
- Step 2: Controlled Feeding and Collection. Animals are fed a measured amount of the experimental diet for a set adaptation period, followed by a total collection period where all feces (and sometimes urine) are quantitatively collected.
- Step 3: Sample Analysis. Feed, feces, and urine are analyzed for mineral content.
- Step 4: Calculation of Apparent Absorption.
Apparent Absorption (%) = [(Mineral ingested - Mineral excreted in feces) / Mineral ingested] * 100
Protocol for Assessing Tissue Mineral Deposition
This terminal assay provides direct evidence of mineral utilization beyond absorption [82].
- Step 1: Tissue Sampling. At the end of a feeding trial, target tissues (e.g., liver, muscle, kidney) are collected, cleaned, and dried.
- Step 2: Acid Digestion. A known weight of the dried tissue is digested with concentrated nitric acid (and potentially hydrogen peroxide) in a controlled environment (hot block or microwave digester) to completely mineralize the organic matrix and release the minerals.
- Step 3: Spectrometric Quantification. The digested sample is diluted and analyzed via ICP-OES or Mass Spectrometry (ICP-MS) to determine the concentration of the target minerals. Results are reported as mg or μg of mineral per gram of tissue dry weight.
Visualization of Chelation and Absorption
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents and Materials for Mineral Chelation Research
| Reagent/Material | Function in Research |
|---|---|
| Amino Acid Ligands (e.g., Glycine, Methionine) | Organic molecules used to synthesize chelates; form stable coordinate covalent bonds with metal ions [81]. |
| Inorganic Mineral Salts (e.g., ZnSO₄, FeSO₄) | Baseline controls for comparative bioavailability studies and starting materials for chelate synthesis [82]. |
| Phytic Acid (Sodium Salt) | Used to create in vitro or in vivo models of mineral antagonism to test the protective efficacy of chelation [23]. |
| Simulated Digestive Fluids (Pepsin, Pancreatin, Bile Salts) | Key components of in vitro simulated gastric and intestinal digestion models to assess mineral bioaccessibility [23]. |
| Stability Constant Analysis Software | Computational tools to model and predict the formation and stability of metal-ligand complexes under varying pH conditions [79]. |
| Analytical Standards (for ICP-OES/MS) | Certified reference materials essential for the accurate quantification of mineral content in feed, tissue, and excreta samples [82]. |
The body of evidence firmly establishes that chelated and organic mineral complexes represent a significant advancement in mineral delivery. Their enhanced stability and targeted absorption mechanisms directly address the core challenge of mineral bioavailability, which is often compromised by dietary inhibitors. The implications extend from improving animal production efficiency and sustainability to informing strategies for combating human micronutrient deficiencies.
Future research will likely focus on refining chelation technology, including the development of customized ligands for specific minerals or physiological targets, and the application of encapsulation techniques for even greater stability [83] [84]. Furthermore, as the global market for chelated minerals continues to grow—projected to reach $6.93 billion by 2029—driven by demand in precision agriculture, sustainable animal husbandry, and human nutraceuticals, the role of rigorous, fundamental research in validating new compounds and their mechanisms becomes ever more critical [83]. Integrating an understanding of mineral chelates is therefore indispensable for any comprehensive research initiative on the inhibitors and enhancers of mineral absorption.
Pharmaceutical formulation optimization represents a critical frontier in advancing modern therapeutics, bridging the gap between drug discovery and clinical application. As we approach 2025, this field is undergoing a profound transformation driven by artificial intelligence, advanced materials, and patient-centric design principles. The global drug formulation market is projected to expand from $1.7 trillion in 2025 to $2.8 trillion by 2035, reflecting a compound annual growth rate of 5.7% [85]. This growth is largely attributable to increasing demands for innovative therapeutics addressing chronic diseases, an aging global population, and more sophisticated drug delivery technologies.
Within this context, understanding inhibitors and enhancers of mineral absorption becomes particularly relevant for formulation scientists. The principles governing mineral bioavailability – including absorption barriers and enhancement strategies – provide valuable paradigms for optimizing drug delivery systems. As pharmaceutical compounds face similar challenges related to solubility, stability, and targeted delivery, lessons from mineral absorption research can inform broader formulation strategies. This whitepaper examines current trends, technologies, and methodologies shaping pharmaceutical formulation optimization, with particular emphasis on timing considerations, dosage form innovation, and combination therapy approaches.
AI-Driven Formulation Optimization
The integration of artificial intelligence (AI) and machine learning (ML) into formulation development is transforming traditional trial-and-error approaches into predictive, data-driven science. AI algorithms can now analyze vast datasets to predict how different chemical compositions will behave under various conditions, significantly reducing development time and resources [86].
From DoE to AI-Driven Design Space Expansion
Traditional Design of Experiment (DoE) approaches, while valuable, explore only a tiny slice of the formulation universe and face "dimensionality explosion" as more parameters are added [87]. AI-driven active learning algorithms represent a paradigm shift, steering successive experimental batches toward the most informative and promising regions of the formulation design space. This dynamic approach enables:
- Multi-objective optimization: Simultaneous evaluation of candidates against full target product profiles, including drug level, stability, release profile, and manufacturability [87]
- Accelerated identification: Surface higher-performing formulations earlier in development, trimming months off the journey to scale-up and market [87]
- Resource reduction: Minimize time and cost by reducing amounts of drug, solvents, and other materials used in development [87]
Leading pharmaceutical companies are increasingly deploying these technologies. Pfizer utilizes AI and machine learning to accelerate formulation development and optimize dosage forms, while Novartis implements digital twins and advanced analytics to improve process understanding and reduce development timelines [85].
Timing Considerations in Formulation Development
AI-driven approaches fundamentally reshape development timing through several mechanisms:
Table 1: Timing Advantages of AI-Driven Formulation
| Development Phase | Traditional Approach | AI-Optimized Approach | Time Savings |
|---|---|---|---|
| Initial Screening | Manual literature review & experimentation | Predictive modeling of chemical interactions | 40-60% |
| Formulation Optimization | Sequential DoE cycles | Parallel multi-parameter optimization | 50-70% |
| Stability Testing | Long-term real-time studies | Predictive stability modeling | 30-50% |
| Scale-up | Empirical adjustments | First-pass success through manufacturability-by-design | 40-60% |
As one industry expert noted: "If you can find a new drug molecule, you can predict its formulation and use our robotic platform to build and test it in the real world" [85]. This highlights the growing integration of in silico prediction with physical validation, creating accelerated development feedback loops.
Advanced Dosage Form Technologies
Dosage form innovation continues to evolve, with oral solid dosages maintaining market leadership at 43.2% share in 2025 due to their convenience, cost-effectiveness, and well-established manufacturing infrastructure [85] [88]. However, significant advances are occurring across all dosage forms to improve efficacy, safety, and patient experience.
Oral Solid Dose Innovations
The oral solid dose (OSD) landscape is witnessing transformative advancements in 2025, particularly in addressing complex challenges related to bioavailability, patient adherence, and personalization:
- High-potency drug formulations: Driven by oncology and specialized therapies, with ongoing innovations in oral formulations for obesity treatments despite challenges in bioavailability and stability [88]
- 3D printing technology: Enables production of multidrug tablets, customized release profiles, and tailored doses, paving the way for personalized medicine [88]
- Patient-centric dosage forms: Innovations including sprinkle formulations, orally disintegrating tablets, and transformative dosage forms ensure better adherence for diverse patient populations including pediatric and geriatric groups [88]
- Enhanced palatability: With over 60% of APIs being inherently bitter, flavoring agents, maskers, and bitterness blockers have become essential tools for improving medication palatability [88]
A notable example of advanced OSD innovation is the Flexipill for hypertension management – a 3D-printed flexible dose combination that addresses polypharmacy challenges while enabling personalization. The formulation incorporates three drugs with distinct release profiles: propranolol HCl as a floating unit achieving 9-hour flotation and over 90% drug release; enalapril maleate printed at 150°C to avoid thermal degradation; and hydrochlorothiazide formulated for immediate release of over 90% of the drug within the first hour [89].
Advanced Drug Delivery Systems
Beyond traditional OSD, sophisticated drug delivery platforms are achieving unprecedented control over therapeutic release and targeting:
Table 2: Advanced Drug Delivery Technologies
| Technology Platform | Key Mechanism | Application Examples | Advantages |
|---|---|---|---|
| Liposomal Systems [90] | Lipid bilayer encapsulation of hydrophilic/hydrophobic drugs | Oncology, antimicrobial therapies | Enhanced pharmacokinetics, reduced side effects, targeted delivery |
| Micro Robotics [91] | Magnetic field-controlled tiny soft robots | Targeted combination therapy with reprogrammable release orders | Site-specific delivery, multiple drug payloads, precision control |
| Extracellular Vesicles [91] | Virus-sized nanoparticles produced by human cells | CRISPR gene-editing delivery, cell therapies | Natural biocompatibility, efficient cell binding and transfer |
| Nanostructured Lipid Carriers (NLCs) [92] | Solid/liquid lipid hybrid nanoparticles | Poorly soluble drugs like edaravone | Improved drug loading, stability, and process control |
These advanced systems share conceptual parallels with mineral absorption enhancers in nature. Just as certain compounds can improve mineral bioavailability in the gastrointestinal tract, these technological approaches enhance drug bioavailability through sophisticated formulation strategies.
Combination Therapy Formulations
Combination therapies present unique formulation challenges, particularly regarding chemical compatibility, release profile coordination, and stability maintenance. Fixed-dose combinations (FDC) have demonstrated effectiveness in improving patient adherence, but they often limit flexibility for dose titration and personalization of treatment [89].
The Polypill Evolution
The concept of combination therapy has evolved significantly from simple fixed-dose formulations:
- First-generation FDCs: Single tablet containing multiple drugs with standard ratios
- Adaptive combinations: Formulations allowing some adjustment through separate components within a single dosage form
- Personalized polypills: 3D-printed combinations tailored to individual patient needs with precise dose titration [89]
The Flexipill approach represents this evolution, creating a flexible dose combination that doesn't require printing at the pharmacy level. It can be printed at a quality-controlled facility and assembled according to patient needs at the point of care [89]. This strategy bridges the gap between regulatory requirements and personalized medicine concepts.
Timing and Release Coordination
Effective combination therapies often require sophisticated release timing to optimize therapeutic effects while minimizing interactions. Advanced formulation strategies enable this through:
- Differential release profiles: Immediate, sustained, and delayed release components within a single dosage form [89]
- Floating systems: Gastric retention mechanisms to improve solubility and bioavailability [89]
- Targeted activation: Environmental triggers such as pH or enzyme activity at specific tissue sites
These approaches mirror principles observed in mineral absorption, where certain "enhancer" compounds create favorable conditions for bioavailability, while "inhibitor" substances must be formulated against to prevent negative interactions.
Experimental Protocols and Methodologies
Formulation Optimization Using DoE
Design of Experiment (DoE) remains a fundamental methodology for systematic formulation development. A representative protocol for lipid-based nanocarrier optimization demonstrates this approach [92]:
Objective: Optimize encapsulation efficiency (EE%) and loading capacity (LC%) of edaravone-loaded nanostructured lipid carriers (NLCs)
Experimental Design:
- Factor Screening: Identify critical formulation parameters (lipid type, solid-to-liquid lipid ratio, drug concentration, processing conditions)
- DoE Matrix: Create structured experimental array varying multiple factors simultaneously
- Response Analysis: Measure EE% and LC% for each experimental condition
- Model Building: Develop mathematical models relating factors to responses
- Optimization: Identify factor combinations predicting optimal performance
- Validation: Confirm model predictions with experimental verification
Key Findings: Lipid type was identified as the most influential formulation parameter, while solid-to-liquid lipid ratio and drug concentration had minor effects [92].
Real-Time Process Monitoring
Advanced analytical technologies now enable real-time monitoring during formulation production:
Integrated SR-DLS Methodology [92]:
- Instrumentation: NanoFlowSizer integrated with Microfluidizer processor
- Measurement Principle: Spatially resolved dynamic light scattering (SR-DLS)
- Implementation: Inline monitoring during high-pressure homogenization
- Applications: Detection of subtle instabilities (micelle formation, particle agglomeration)
- Advantages: Early detection of formulation issues, superior to conventional offline DLS
This methodology exemplifies the trend toward continuous manufacturing and real-time quality assurance, significantly reducing traditional batch-to-batch variability.
Visualization of Formulation Workflows
AI-Driven Formulation Optimization Pathway
AI-Driven Formulation Workflow: This diagram illustrates the integrated, data-driven approach to modern formulation development, highlighting the continuous feedback between design, testing, and optimization phases enabled by AI and real-time monitoring technologies.
Mineral Absorption Principles in Formulation Design
Mineral Absorption Informing Formulation: This visualization demonstrates how principles from mineral absorption research – specifically the balance between enhancers and inhibitors – provide valuable conceptual frameworks for pharmaceutical formulation strategies aimed at optimizing bioavailability.
Research Reagent Solutions Toolkit
Table 3: Essential Formulation Research Reagents and Technologies
| Reagent/Technology | Function in Formulation | Application Examples | Key Characteristics |
|---|---|---|---|
| Precirol ATO 5 [92] | Solid lipid for NLCs | Poorly soluble drug encapsulation | High melting point, biocompatible, sustained release properties |
| Gelucire 43/01 [92] | Solid lipid for NLCs | Temperature-sensitive formulations | Low melting point, self-emulsifying properties |
| Labrafac Lipophile WL 1349 [92] | Liquid lipid for NLCs/NEs | Bioavailability enhancement | Medium-chain triglycerides, excellent solvent properties |
| Nitrite Scavengers (e.g., Ascorbic Acid) [88] | Mitigate nitrosamine formation | Reformulation for compliance | Blocks nitrosation reactions, regulatory compliance |
| Bitterness Blockers/Maskers [88] | Improve palatability | Pediatric/geriatric formulations | Taste masking without affecting drug release |
| Chitosan-TPP Nanoparticles [90] | Mucoadhesive drug carrier | Hormone delivery (veterinary applications) | Biocompatible, biodegradable, positive surface charge |
| Rotaxane Molecular Devices [91] | Force-controlled release | Targeted delivery to injury/tumor sites | Mechanophoric mechanism, multiple payload capacity |
Pharmaceutical formulation optimization in 2025 represents a sophisticated integration of AI-driven prediction, advanced materials science, and patient-centric design principles. The field has evolved from traditional trial-and-error approaches to data-rich, model-guided development processes that simultaneously optimize multiple formulation parameters. Timing considerations have been fundamentally reshaped through predictive modeling and real-time monitoring technologies, while dosage forms have advanced toward personalized, adaptable systems that balance therapeutic efficacy with patient experience.
The conceptual framework of mineral absorption research – with its focus on enhancers and inhibitors of bioavailability – provides valuable parallels for pharmaceutical scientists developing advanced formulation strategies. Just as mineral enhancers improve nutrient uptake, advanced formulation technologies enhance drug delivery through nanoscale engineering, targeted release systems, and bioavailability optimization.
As the field progresses, the integration of continuous manufacturing, digital twins, and AI-guided development will further accelerate formulation timelines while improving product quality. The convergence of these technologies points toward a future of truly personalized medicines – formulations tailored not just to disease states but to individual patient physiology, preferences, and needs. For researchers and drug development professionals, mastering these evolving tools and methodologies will be essential for developing the next generation of advanced therapeutics.
The efficacy of mineral supplementation is not solely determined by the administered dose but by its bioavailability—the proportion that is absorbed, transported, and utilized in physiological processes. This bioavailability is influenced by a complex interplay of dietary factors, host physiology, and life stage. Research into the inhibitors and enhancers of mineral absorption is thus critical for developing effective nutritional interventions. This is particularly true for special populations—the elderly, individuals with inflammatory conditions, and post-surgical patients—who face unique physiological challenges that can profoundly alter their nutritional requirements and absorption capabilities. This whitepaper provides an in-depth technical guide to the formulation of mineral supplements tailored to the distinct needs of these groups, framed within the broader context of mineral absorption research.
Mineral Absorption: Core Principles and Research Constructs
A foundational understanding of mineral absorption mechanics is a prerequisite for developing targeted formulations.
Fundamentals of Absorption Pathways
Iron absorption serves as a paradigm for the tight regulation of mineral uptake, particularly for non-heme iron, which is highly susceptible to dietary factors [93]. The process is initiated in the duodenum and proximal jejunum, where the acidic gastric environment facilitates the reduction of insoluble ferric iron (Fe³⁺) to absorbable ferrous iron (Fe²⁺) via the brush border enzyme duodenal cytochrome B (Dcytb) [93]. The divalent metal transporter 1 (DMT1) then mediates the uptake of Fe²⁺ across the apical membrane of the enterocyte. Intracellular iron is either stored as ferritin or exported into circulation via ferroportin, the sole known iron exporter [93]. The hormone hepcidin serves as the master regulator of systemic iron homeostasis; elevated hepcidin levels, often seen in inflammatory states, trigger the internalization and degradation of ferroportin, thereby sequestering iron in enterocytes and macrophages and reducing plasma iron availability [93].
The following diagram illustrates the key cellular and molecular players in iron absorption and its regulation.
Key Bioavailability Concepts and Assessment Methodologies
Bioavailability is defined as the proportion of an ingested nutrient that is available for use in normal physiological functions and storage [18]. Key methodologies for its assessment include:
- Balance Studies: Measuring the difference between nutrient ingestion and excretion. This provides an estimate of apparent absorption [18].
- Ileal Digestibility: A more precise method that measures the difference between intake and the nutrient remaining in ileal contents, avoiding potential confounding from colonic microbial activity [18].
- Stable Isotope Tracers: The use of isotopically labeled minerals (e.g., ⁵⁷Fe, ⁶⁷Zn) allows for the precise tracking of absorption, metabolism, and distribution within the body without the ethical concerns of radioisotopes.
- Systemic Biomarker Response: Monitoring changes in functional biomarkers (e.g., serum ferritin for iron, erythrocyte superoxide dismutase for copper) in response to supplementation provides an integrated measure of bioavailability and utilization.
Special Population Profiles and Formulation Strategies
The generic "one-size-fits-all" approach to mineral supplementation is inadequate for populations with altered physiology. The table below summarizes the core challenges and targeted formulation strategies for the populations in focus.
Table 1: Special Population Profiles and Targeted Formulation Strategies
| Population | Core Physiological Challenge | Key Mineral Concerns | Targeted Formulation Strategy |
|---|---|---|---|
| Elderly | Inflammaging: Chronic low-grade inflammation; elevated pro-inflammatory cytokines (IL-6, TNF-α, CRP) [94].Achlorhydria: Reduced stomach acid, impairing iron and calcium solubility [93].Dysbiosis: Altered gut microbiota affecting mineral absorption [95]. | Iron: Hepcidin-mediated sequestration [93].Calcium & Vitamin D: Critical for bone health; absorption often impaired [96].Zinc & Selenium: Crucial for immune function; deficiencies common [94]. | • Anti-inflammatory combinations: Co-supplementation with Omega-3 PUFAs (1000 mg EPA+DHA) to reduce inflammation and improve mineral utilization [97].• Chelated Minerals: Use of bisglycinate or citrate forms to bypass need for stomach acid.• Probiotic Co-administration: Selected strains (e.g., Lactobacillus, Bifidobacterium) to improve mineral bioaccessibility [95]. |
| Inflammatory Conditions | Hepcidin Upregulation: Inflammatory cytokines (e.g., IL-6) stimulate hepcidin production, blocking iron export via ferroportin, leading to "Anemia of Chronic Disease" [93]. | Iron: Central to the pathology of anemia of inflammation. | • Hepcidin Antagonists: Research into compounds that suppress hepcidin expression.• Ascorbic Acid: High-dose vitamin C (molar ratio 2:1 to 4:1 AA:Iron) to enhance non-heme iron absorption, potentially countering dietary inhibitors [98].• Siderophore Mimetics: Development of novel chelators that can shuttle iron into circulation despite inflammatory block. |
| Post-Surgical (Gastrectomy) | Anatomical Alteration: Bypass of duodenum/proximal jejunum (primary sites of iron/calcium absorption); loss of stomach acid and intrinsic factor [96].Rapid Transit & Malabsorption [96]. | Iron, Calcium, Vitamin B₁₂, Vitamin D, Zinc: High risk of severe deficiencies due to combined restrictive and malabsorptive physiology [96]. | • High-Potency, Specialized Formulations: Use of bariatric-style multivitamins meeting ASMBS guidelines (e.g., ≥45 mg elemental iron, 1000 µg B₁₂) [96].• Chemical Form Selection: Use of calcium citrate (acid-independent) over carbonate; liquid or chewable forms for better tolerance.• Timed-Dose Separation: Administering calcium and iron supplements separately to avoid competitive inhibition [96]. |
Experimental Protocols for Key Research Areas
Protocol 1: Assessing the Efficacy of an Iron Absorption Enhancer in an Inflamed Model
- Objective: To determine if a candidate enhancer (e.g., ascorbic acid) can overcome hepcidin-mediated iron blockade in a model of inflammation.
- Cell Culture: Utilize Caco-2 cell monolayers as an in vitro model of the intestinal epithelium.
- Inflammatory Priming: Pre-treat cells with IL-6 (e.g., 10 ng/mL for 24 hours) to upregulate hepcidin expression.
- Intervention: Apply ferric ammonium citrate as an iron source with varying concentrations of ascorbic acid (e.g., molar ratios from 0.5:1 to 4:1: AA:Iron).
- Outcome Measures:
- Transepithelial Transport: Quantify ferritin expression in cells (storage) and iron content in the basolateral compartment (absorption) using ICP-MS.
- Gene Expression: Measure mRNA levels of hepcidin, DMT1, and ferroportin via qPCR.
- Protein Validation: Confirm ferroportin protein levels via Western blot.
Protocol 2: Evaluating a Multi-Micronutrient Formulation in an Aged Cohort
- Study Design: Randomized, double-blind, placebo-controlled 12-week trial, following the model of Hahn et al. [97].
- Participants: Healthy adults aged ≥70 years.
- Intervention Group: Daily supplement containing:
- Vitamin D (50 µg cholecalciferol)
- Omega-3 PUFAs (1000 mg EPA+DHA)
- B-Vitamins (400 µg folic acid, 100 µg cobalamin)
- Zinc (e.g., 15-20 mg)
- Selenium (e.g., 100 µg)
- Control Group: Placebo.
- Primary Outcome: Change in aggregated inflammatory (INFLA) score comprising CRP, IL-6, and other biomarkers [97].
- Secondary Outcomes: Changes in mineral status biomarkers (e.g., serum ferritin, zinc), Omega-3 Index, and functional measures (e.g., muscle strength).
Pathway Visualization and Research Toolkit
Nutrient Modulation of Inflammaging
The following diagram synthesizes the mechanistic pathways through which targeted nutrients modulate the chronic low-grade inflammation characteristic of aging.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Models for Investigating Mineral Absorption
| Reagent / Model | Function / Rationale | Example Application |
|---|---|---|
| Caco-2 Cell Line | Human colorectal adenocarcinoma cell line that, upon differentiation, forms polarized monolayers with brush border enzymes and transporters, mimicking intestinal epithelium. | The standard in vitro model for studying transepithelial transport and mechanism of mineral absorption and interaction with enhancers/inhibitors [98]. |
| Stable Isotopes | Non-radioactive isotopes (e.g., ⁵⁷Fe, ⁷⁰Zn, ⁴⁴Ca) used to trace the metabolic fate of minerals from a specific dose within the body. | Allows for precise measurement of mineral absorption from a test meal or supplement in human clinical trials, without confounding by endogenous mineral pools. |
| IL-6 Cytokine | A key pro-inflammatory cytokine that upregulates the expression of the iron-regulatory hormone hepcidin. | Used in vitro (Caco-2) or in vivo to induce an "anemia of inflammation" phenotype and test the efficacy of interventions aimed at overcoming hepcidin blockade [93]. |
| Dcytb & DMT1 Assays | Enzymatic and transporter activity assays. Dcytb reduces Fe³⁺ to Fe²⁺, while DMT1 transports Fe²⁺ across the apical membrane. | Quantifying the impact of genetic polymorphisms, nutritional status, or pharmaceutical agents on the initial critical steps of iron absorption [93]. |
| Third-Party Tested Supplements (e.g., NSF Certified) | Ensures supplement composition matches label claims and is free from contaminants. Critical for clinical research reproducibility and safety. | Used as the verified intervention in human trials to ensure results are attributable to the stated ingredients at the declared doses [99] [96]. |
The paradigm of mineral supplementation is shifting from generic potency to targeted bioavailability. Addressing the needs of special populations requires a deep understanding of their unique pathophysiology—specifically, the inflammatory milieu in the elderly, hepcidin dysregulation in chronic disease, and anatomical disruption in post-surgical patients. The formulation strategies outlined herein, including the use of specific chemical forms, anti-inflammatory adjuvants, and high-potency specialized blends, represent a scientifically-grounded approach to bypassing these barriers. Future research must focus on further elucidating the molecular pathways governing mineral metabolism in these populations, validating the efficacy of targeted formulations in robust clinical trials, and exploring novel enhancers like specific probiotic strains. By integrating precise nutrition with clinical medicine, we can develop more effective interventions that improve not only biochemical status but also functional health outcomes.
Critical Evaluation of Enhancement Technologies and Clinical Translation
The efficacy of mineral supplements is fundamentally governed by their bioavailability—the proportion of an ingested nutrient that is absorbed, transported to tissues, and utilized in physiological functions [18]. Within pharmaceutical and nutraceutical development, a central challenge lies in overcoming the inherent low bioavailability of many essential minerals, which is influenced by the mineral's chemical form, the presence of dietary inhibitors, and host-specific factors [100] [18]. This analysis provides a comparative evaluation of organic versus inorganic minerals and examines the scientific evidence for various platforms designed to enhance their absorption. Framed within the broader context of mineral absorption research, this review synthesizes data on bioavailability, explores the mechanisms of absorption enhancers, and details advanced experimental methodologies, serving as a technical guide for researchers and drug development professionals.
Fundamental Concepts: Mineral Forms and Bioavailability
Minerals are essential inorganic elements, but in nutritional and pharmaceutical contexts, they are delivered as compounds categorized by their chemical structure into organic and inorganic forms.
- Inorganic Minerals: These are simple compounds where the mineral is bound to an inorganic ligand, such as oxides, sulfates, chlorides, or carbonates [101]. Examples include magnesium oxide and ferrous sulfate. Their bioavailability is often limited because they can ionize readily in the gastrointestinal tract, making them susceptible to interactions with dietary inhibitors and leading to poor absorption and potential gastrointestinal distress [25] [101].
- Organic Minerals: This category encompasses minerals bound to carbon-based molecules. A critical subset is chelated minerals, where the mineral ion is coordinatively bound to an organic ligand, such as an amino acid (e.g., bisglycinate) or a protein (e.g., proteinate) [25] [101]. The chelation process, inspired by mineral binding in nature (e.g., magnesium in chlorophyll), forms a stable, ring-like structure that protects the mineral from antagonists in the gut and facilitates its absorption via pathways designed for organic molecules [25].
The concept of bioaccessibility is a critical prerequisite to bioavailability, referring to the fraction of the mineral that is released from the food or supplement matrix and becomes soluble in the gastrointestinal lumen, making it available for absorption [102] [18].
Comparative Efficacy of Organic and Inorganic Minerals
Extensive research, summarized in the table below, demonstrates that organic minerals, particularly chelates, consistently offer superior bioavailability and functional outcomes compared to their inorganic counterparts.
Table 1: Comparative Bioavailability and Efficacy of Organic vs. Inorganic Minerals
| Mineral & Form | Study Model | Key Comparative Findings | Mechanistic Insights |
|---|---|---|---|
| IronFerrous Bisglycinate vs. Ferrous Sulfate | Human Clinical Trials [25] | Absorption was up to 5x more effective in the presence of phytates. WHO designated it a "fortificant of choice" [25]. | The chelate structure shields the iron ion from phytates, preventing the formation of insoluble complexes [25]. |
| IronFood Processing | In Vitro Digestion Model [103] | Roasting increased iron bioaccessibility in oilseeds by 30-80%; germination in millets by 5-10%; blanching in leafy greens by ~200% [103]. | Processing techniques (roasting, germination, blanching) significantly reduce antinutrient content (phytates, tannins) [103]. |
| CalciumCalcium Bisglycinate vs. Carbonate/Citrate | Human Clinical Trials [25] | Calcium Bisglycinate delivered up to 2x the absorption of calcium citrate and carbonate [25]. | The chelate's neutral charge and high solubility enhance absorption efficiency and improve tolerability [25]. |
| Zinc & ManganeseOrganic Proteinate vs. Inorganic Sulfates | Broiler Study [101] | Birds fed proteinate minerals showed superior growth performance, higher meat yield, and improved immunity [101]. | Organic minerals are highly bioavailable and require lower dietary concentrations, reducing mineral excretion and environmental impact [101]. |
| Multi-MineralPowder (AG1) vs. Tablet MVM | In Vitro SHIME Model [102] | The powder format showed significantly higher (p < 0.05) bioaccessibility and bioavailability of Mg, Ca, and Zn during small intestine simulation [102]. | Powdered formulations forego the disintegration requirement of tablets, allowing for more immediate and complete mineral release [102]. |
The superior performance of organic minerals is attributed to several key mechanisms. They are protected from potent dietary inhibitors like phytates and oxalates, which form insoluble complexes with free mineral ions, rendering them unabsorbable [25] [103]. Furthermore, their structure often allows them to be absorbed via amino acid or peptide transporters in the intestine, which are more efficient than the passive diffusion pathways typically used by inorganic ions [25]. This efficient absorption also translates to better gastrointestinal tolerability, as evidenced by a 64% reduction in adverse events for Ferrous Bisglycinate compared to other iron salts [25].
Platforms and Technologies for Enhancing Mineral Absorption
Beyond the intrinsic form of the mineral, several platforms and technologies can be employed to enhance absorption.
Absorption Enhancers (Bioenhancers)
These are compounds that temporarily and reversibly improve the permeability of the intestinal membrane or inhibit metabolic processes that degrade the active compound [104]. They are particularly relevant for overcoming the barriers to macromolecule and mineral absorption.
- Chemical Permeation Enhancers: These include surfactants and bile salts that alter membrane fluidity. However, issues with reproducibility and safety concerns regarding long-term use have limited their application [104].
- Herbal Bioenhancers: Derived from plants, these are often deemed safer and have demonstrated significant efficacy.
- Piperine (from black pepper): A well-researched bioenhancer that acts through multiple mechanisms, including inhibition of drug-metabolizing enzymes (e.g., CYP3A4) and efflux transporters (P-glycoprotein), as well as slowing gastrointestinal transit to increase absorption time [104].
- Aloe Vera, Ginger, and Caraway Extracts: These have shown clinical promise in enhancing the bioavailability of various drugs and nutrients, such as vitamins C and E [104].
- Glycyrrhizin (from licorice): Shown to enhance the intestinal absorption of drugs by increasing membrane permeability [104].
- Lysergol (from morning glory species): Patented as a bioenhancer for antibiotics and other drugs [104].
Delivery System Technologies
The format and composition of the delivery system itself are critical for mineral bioavailability.
- Powder vs. Tablet Delivery: As demonstrated in the SHIME model study, a powdered multivitamin and mineral (MVM) supplement demonstrated significantly higher mineral bioaccessibility and bioavailability than an identical formulation in a tablet. The tablet's requirement for disintegration in the GI tract is a major rate-limiting step, whereas powders are immediately available for dissolution [102].
- Synbiotic Formulations: Combining minerals with prebiotics and probiotics (synbiotics) can enhance bioavailability. A healthy gut microbiota can increase the absorption of vitamins and minerals, and specific strains may help degrade antinutrients like phytates [18].
- Microencapsulation and Lipid-Based Formulations: These technologies protect minerals from degradation in the GI tract and can facilitate absorption, particularly for fat-soluble compounds [18].
Food Processing Techniques
Traditional and sequential food processing methods are effective, low-tech strategies for improving mineral bioaccessibility from food sources.
- Sequential Processing: Combining techniques like soaking, germination, and roasting can lead to a dramatic reduction (19-92%) in phytate and tannin content, thereby liberating bound minerals [103].
- Thermal Processing: Roasting and blanching disrupt plant cell walls and degrade antinutrients, significantly enhancing iron bioaccessibility [103].
- Germination (Sprouting): Activates endogenous phytase enzymes that break down phytic acid, reducing its mineral-chelating power [103].
Experimental Models for Assessing Bioavailability
Selecting appropriate experimental models is crucial for the accurate evaluation of mineral bioavailability in research and development.
Table 2: Experimental Models for Assessing Mineral Bioavailability
| Model Type | Description | Key Applications | Example |
|---|---|---|---|
| In Vitro Digestion Models | Simulates human digestion (stomach, small intestine) in a bioreactor. | Measures bioaccessibility—the fraction of mineral solubilized during digestion. | SHIME (Simulator of the Human Intestinal Microbial Ecosystem): Used to compare powder vs. tablet MVM supplements, measuring mineral concentration in digested samples and dialysate [102]. |
| Cell Culture Models | Uses human intestinal cell lines (e.g., Caco-2) grown on permeable supports. | Studies intestinal transport and uptake mechanisms of minerals. | Used to investigate the transport pathways of mineral chelates (e.g., amino acid-mediated) and the effect of permeation enhancers [100]. |
| Animal Models | Studies in live animals (e.g., rodents, broiler chickens). | Assesses growth, tissue mineral deposition, and physiological outcomes. | Broiler chicken studies comparing effects of organic and inorganic trace minerals on growth performance, carcass traits, and immunity [101]. |
| Human Clinical Trials | Balance studies, ileal digestibility measurements, or pharmacokinetic studies in human subjects. | Provides the most relevant data on absolute and relative bioavailability for a target population. | Balance studies measuring the difference between mineral ingestion and excretion; used to validate findings from other models [18]. |
Detailed Experimental Protocol: In Vitro SHIME Model
The following workflow outlines a standardized protocol for assessing mineral bioaccessibility and bioavailability using the SHIME model, a method used in recent research [102].
Title: SHIME Mineral Bioaccessibility Workflow
Protocol Steps:
- Initial Soluble Fraction Quantification: The test products (tablet and powder) are first homogenized in water. The soluble mineral content is quantified using Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES) to establish a baseline [102].
- SHIME Inoculation and Operation: The tablet (ingested whole) and powder are introduced into the SHIME bioreactor, which maintains physiological conditions (pH, temperature, mixing) and simulates the digestive secretions of the stomach and duodenum [102].
- Bioaccessibility Measurement: Samples are taken at the end of the simulated stomach and duodenum phases. The mineral content in these samples represents the bioaccessible fraction—the amount released from the matrix and available for absorption [102].
- Bioavailability Assessment: The digested material is passed through a dialysis membrane with a molecular weight cut-off that mimics the intestinal barrier. The minerals that pass through this membrane into the dialysate over time (e.g., at 1, 2, and 3 hours) represent the bioavailable fraction—the portion that is absorbable [102].
- Data Analysis: Mineral concentrations from all phases are analyzed via ICP-OES. Statistical comparisons (e.g., ANOVA) are made between the test products to determine significant differences in bioaccessibility and bioavailability [102].
The Scientist's Toolkit: Key Research Reagents and Materials
This section details essential materials and reagents used in the experiments cited throughout this analysis, providing a practical resource for experimental design.
Table 3: Essential Research Reagents and Materials for Mineral Absorption Studies
| Reagent / Material | Function in Research | Application Example |
|---|---|---|
| Mineral Amino Acid Chelates(e.g., Bisglycinate) | Organic mineral standard; ligand protects mineral from antagonists and facilitates amino acid transport. | Used as the active intervention in comparative studies against inorganic salts (e.g., sulfates, oxides) to demonstrate enhanced bioavailability [25] [101]. |
| Phytic Acid (Sodium Salt) | A potent dietary antagonist; used to create a controlled challenge in absorption models. | Added to in vitro digestion models or animal diets to simulate a high-phytate meal and test the resilience of different mineral forms [25] [103]. |
| Clinoptilolite | A natural zeolite with high cation-exchange capacity; used as a detoxifying agent and potential mineral carrier. | Studied in veterinary and human medicine for its ability to bind ammonia and toxins; its porous structure is explored for drug and nutrient delivery [105]. |
| Piperine (≥97% purity) | A gold-standard herbal bioenhancer; inhibits metabolism and efflux transporters. | Used as a positive control in studies investigating the enhancement of bioavailability for drugs and nutrients [104]. |
| SHIME Reactor System | A dynamic, computer-controlled in vitro model of the human gastrointestinal tract. | Used to simulate the entire human digestion process, allowing for continuous monitoring of mineral bioaccessibility and bioavailability under physiologically relevant conditions [102]. |
| ICP-OES/Mass Spectrometry | Analytical technique for precise quantification of mineral elements in complex samples (digesta, dialysate, tissues). | The primary method for measuring mineral concentration in samples from in vitro and in vivo studies to calculate absorption and retention [102]. |
| Caco-2 Cell Line | A human colon adenocarcinoma cell line that differentiates into enterocyte-like cells. | A standard in vitro model for studying intestinal permeability and transport mechanisms of minerals and drugs [100]. |
This comparative analysis establishes that the chemical form and delivery platform of a mineral are decisive factors in its efficacy. Organic minerals, particularly chelates like bisglycinates and proteinates, demonstrate consistently superior bioavailability and functional benefits over inorganic salts, primarily due to their resistance to dietary inhibitors and efficient absorption pathways. The strategic use of absorption enhancers like piperine, advanced delivery systems such as synbiotic powders, and optimized food processing techniques provide a multi-faceted toolkit for significantly improving mineral absorption. For researchers and drug development professionals, the rigorous application of advanced models like the SHIME system and Caco-2 cells is critical for the accurate evaluation of new mineral formulations. The convergence of these strategies—selecting highly bioavailable organic forms, incorporating safe and effective bioenhancers, and employing optimal delivery systems—represents the future frontier for developing next-generation mineral supplements and pharmaceuticals to effectively address global micronutrient deficiencies.
Within the broader research on inhibitors and enhancers of mineral absorption, this review synthesizes clinical evidence on three strategic interventions: probiotic supplementation, permeation-enhancing technologies, and food fortification. Minerals are vital for numerous physiological processes, and their deficiency presents a significant global health challenge. The efficacy of mineral absorption is governed by a complex interplay of dietary factors, gut environment, and intestinal permeability. This clinical evidence review examines findings from human trials to evaluate the real-world impact and methodological approaches of these interventions, providing a technical guide for researchers and drug development professionals.
Probiotics and Mineral Absorption: Clinical Evidence and Mechanisms
Human Clinical Trial Evidence
Probiotics, defined as live microorganisms which when administered in adequate amounts confer a health benefit on the host, have garnered significant interest for their potential to modulate mineral absorption. A recent systematic review analyzed 31 clinical trials with a total of 4,611 participants, focusing on the effects of probiotics, prebiotics, and synbiotics on heavy metal and essential mineral status [106]. Among these trials, 23 investigated probiotics, five looked at prebiotics, and three explored synbiotics, with probiotic doses ranging from 10^7 to 2.5 × 10^10 CFU/day [106].
Table 1: Summary of Key Clinical Findings on Probiotics and Mineral Absorption
| Mineral / Health Outcome | Probiotic Strain / Intervention | Clinical Findings | Study Details |
|---|---|---|---|
| Iron Absorption | Prebiotic galactooligosaccharides | Enhanced iron absorption in women and children [106] | Significant improvement in target populations |
| Iron Status | Lactobacillus plantarum | Significantly increased nonheme dietary iron absorption [106] | Meta-analysis confirmation |
| Diarrhea Treatment | Probiotics combined with Zinc | Significantly reduced time to resolution of vomiting and diarrhea vs. zinc alone [106] | Improved treatment efficacy for antibiotic-associated diarrhea |
| Heavy Metal Detoxification | Various Probiotics (e.g., L. plantarum) | Decrease in toxic metal levels in pregnant women; no similar effects observed in children [106] | Contingent on population and specific metal |
| General Mineral Absorption | Various Probiotics | Modulation of mineral absorption and mitigation of deficiencies [95] | Strain-specific and dose-dependent effects |
The mechanisms by which probiotics enhance mineral absorption are multifaceted. Lactobacillus plantarum is thought to positively affect dietary nonheme iron absorption through several mechanisms: (1) the production of p‐hydroxyphenyllactic acid, a microbial by‐product that can enhance the conversion of ferric iron to the more readily available ferrous form; (2) enhanced iron uptake into enterocytes through increased mucin production; and (3) immunomodulation through the suppression of hepcidin production, resulting in increased iron bioavailability [106]. Furthermore, probiotics can promote mineral absorption by reducing the pH in the gut lumen through the production of short-chain fatty acids (SCFAs), which can help solubilize minerals and make them more available for absorption [95].
Experimental Protocols in Probiotic-Mineral Research
Typical human trial protocols involve randomized, controlled designs where participants are assigned to receive either the probiotic intervention or a matched placebo for a defined period. For instance, trials investigating iron absorption often employ stable isotope techniques to precisely measure fractional iron absorption from test meals with and without probiotic co-administration. Baseline and post-intervention measurements of mineral status (e.g., serum mineral levels, ferritin for iron stores) are standard, alongside analysis of gut microbiota composition to correlate changes with clinical outcomes.
A key methodological consideration is the standardization of the probiotic intervention, including the specific strain(s), dosage (CFU/day), formulation, and delivery matrix (e.g., dairy product, capsule), all of which can influence viability and efficacy. The composition of an individual's gut microbiota is a significant variable, posing a challenge for establishing universal dosage guidelines [95].
Permeation Enhancers in Drug Delivery: Translational Insights
Chemical Penetration Enhancers for Macromolecules
Permeation enhancers are functional excipients included in formulations to increase absorption by enhancing membrane permeation, particularly for compounds with poor inherent permeability [44]. This technology is crucial for developing non-injection formulations for peptides, proteins, and other macromolecules. While much research focuses on transdermal delivery, the principles are informative for gastrointestinal absorption challenges.
Table 2: Classes and Examples of Chemical Permeation Enhancers
| Class of Enhancer | Representative Examples | Postulated Mechanism of Action |
|---|---|---|
| Surfactants | Sodium lauryl sulfate (SLS), Polyoxyethylene ethers, Tween [107] | Disorganization of membrane architecture; loosening of tight junctions [107] |
| Fatty Acids & Derivatives | Oleic acid, Caprylic acid, Acylcarnitines, Mono/diglycerides [107] | Fluidization of lipid bilayers to ease transcellular transport [107] |
| Bile Salts | Sodium glycocholate, Sodium taurocholate, Sodium deoxycholate [107] | Dissociation of drug complexes; membrane disruption [107] |
| Chelating Agents | EDTA, Citric acid [107] | Complexation of calcium/magnesium to disrupt tight junctions [107] |
| Cyclodextrins | Various derivatives [107] | Improved solubility and potential membrane interaction |
The primary barrier to transdermal delivery, including for minerals, is the stratum corneum (SC), the outermost 10–15 µm thick layer of the epidermis [108]. Permeation enhancers such as ionic liquids (ILs), nanoparticles, and peptides are designed to temporarily disrupt this tightly organized barrier, often by interacting with the lipid bilayer to facilitate diffusion [108]. For oral delivery of peptides like insulin, permeation enhancers must overcome intestinal epithelial barriers, often by transiently opening paracellular pathways or facilitating transcellular transport [44].
Methodological Considerations in Permeation Enhancement Studies
Clinical trials for permeation enhancers, especially those intended for systemic delivery, require careful design to establish both efficacy and safety. These studies often involve pharmacokinetic assessments to measure the increased bioavailability of the co-administered active drug (e.g., insulin, heparin) compared to a control. Safety monitoring is paramount, particularly for chronic use, to assess potential local irritation or damage to the mucosal barrier [107] [44].
For example, a trial might investigate an oral insulin capsule containing a permeation enhancer versus subcutaneous insulin injection in diabetic patients. Key endpoints would include pharmacodynamic measures like postprandial glucose control, alongside pharmacokinetic parameters (AUC, C~max~) to quantify the relative bioavailability enabled by the enhancer. Skin irritation tests (e.g., patch testing) are standard for transdermal formulations [108].
Fortified Foods and Mineral Bioavailability: Public Health Impact
Clinical Evidence and Technological Innovations
Food fortification, the practice of adding essential micronutrients to staple foods, is a well-established public health strategy to combat global micronutrient deficiencies [109]. The clinical impact of fortification is significant, with programs for iodized salt, iron-fortified flour, and vitamin D-fortified milk demonstrating success in reducing disorders like goiter, anemia, and rickets [110] [109].
A major challenge in mineral fortification is the presence of dietary inhibitors, such as phytic acid in cereals, which strongly chelates minerals like iron and zinc, drastically reducing their bioavailability [23]. Research has therefore focused on developing processing methods to mitigate these effects. Fermentation and germination have emerged as promising methods for activating endogenous wheat phytases, which effectively hydrolyze phytic acid [23]. Similarly, the addition of exogenous microbial phytases during food processing demonstrates significant potential for enhancing mineral absorption from plant-based foods [23].
Emerging technologies are further advancing the field. Nano-encapsulation can protect minerals from degradation, improve stability, and potentially enhance delivery efficiency [110]. Genetic biofortification, exemplified by "Golden Rice," aims to increase the intrinsic mineral content of crops through agronomic and genetic innovations [109]. These strategies are complemented by novel delivery systems such as 3D food printing and edible coatings, which aim to improve nutrient stability and consumer acceptance [110].
Protocols for Assessing Efficacy of Fortified Foods
Human trials for fortified foods often deploy randomized controlled trials (RCTs) or controlled dietary interventions. These studies typically recruit specific at-risk populations (e.g., children, pregnant women) and provide them with the fortified food product or a non-fortified control over weeks or months.
Key outcome measures include changes in serum or plasma levels of the target mineral, functional biomarkers (e.g., hemoglobin for iron, specific enzymes for selenium), and clinical assessment of deficiency-related symptoms. For iron, stable isotope studies are the gold standard for measuring absorption efficacy directly. Bioaccessibility tests, which simulate human digestion in vitro, are also widely used as a preliminary screening tool for new fortificants and formulations [23]. The choice of the mineral compound (e.g., ferrous sulfate vs. sodium iron EDTA for iron) is critical, as it greatly influences bioavailability, cost, and sensory properties of the final food product.
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Reagents and Materials for Mineral Absorption Research
| Reagent / Material | Function in Research | Application Example |
|---|---|---|
| Specific Probiotic Strains (e.g., Lactobacillus plantarum, L. acidophilus) | Direct intervention to modulate gut environment for improved mineral solubility and uptake. | Human trials on iron absorption; heavy metal detoxification studies [106] [95]. |
| Exogenous Microbial Phytases | Enzymatic degradation of phytic acid (a key mineral absorption inhibitor) in food matrices. | Pre-treatment of cereals/grains to improve bioaccessibility of iron and zinc [23]. |
| Chemical Permeation Enhancers (e.g., Sodium Caprate, Chitosan) | Transiently increase paracellular or transcellular permeability of intestinal epithelium. | Formulation component in oral delivery studies for macromolecules (e.g., peptides) [44]. |
| Stable Isotopes (e.g., ⁵⁷Fe, ⁶⁷Zn) | Precise, quantitative tracking of mineral absorption and metabolism in humans. | Gold-standard method for measuring fractional absorption of minerals from fortified foods [23]. |
| Encapsulation Materials (e.g., polysaccharides, lipids for nano/micro-encapsulation) | Protect reactive minerals from oxidation/inhibitors and control release in the gut. | Developing next-generation fortificants for sensitive minerals like iron [110]. |
This clinical evidence review demonstrates that probiotics, permeation enhancers, and fortified foods represent three distinct yet potentially complementary strategies for overcoming the key challenges of mineral absorption. Human trials confirm that specific probiotic strains can significantly enhance the absorption of essential minerals like iron, primarily through mechanisms that improve solubility and the gut's absorptive capacity. Permeation enhancers, while more advanced in drug delivery for macromolecules, offer a promising technological principle for improving the bioavailability of mineral supplements. Fortified foods remain a foundational public health tool, with ongoing innovations in processing and delivery systems actively addressing the persistent problem of mineral bioavailability. The future of this field lies in the continued refinement of these interventions through rigorous, well-designed human trials, and potentially, in their strategic combination to achieve synergistic benefits for global nutritional health.
Within the critical field of mineral absorption research, the role of pharmaceutical excipients is often underestimated. These components, traditionally viewed as inert, are now recognized as potent modulators of bioavailability, capable of acting as both enhancers and inhibitors of nutrient uptake. Their interaction with the gastrointestinal environment, particularly its mucosal integrity, presents a complex landscape of efficacy and safety considerations that researchers and drug development professionals must navigate. This whitepaper provides an in-depth technical examination of the safety and regulatory considerations for excipients, with a specific focus on their impact on mucosal integrity, systemic toxicity profiles, and the strategic selection of approval pathways. Framed within the broader thesis of mineral absorption research, this guide synthesizes current experimental data and regulatory frameworks to inform the development of safer and more effective mineral-based therapeutics and supplements. The following sections will detail specific excipients' detrimental effects, outline relevant experimental methodologies for safety assessment, and provide a strategic overview of the regulatory landscape.
Impact of Excipients on Mucosal Integrity and Toxicity Profiles
Excipients can significantly compromise intestinal barrier function and induce toxicological effects, as demonstrated by rigorous in vivo studies. The widely used pharmaceutical excipient PEG400 serves as a pertinent case study.
Experimental Findings on PEG400
A 2023 study investigated the effects of 14-day PEG400 administration on healthy mice, revealing significant adverse outcomes related to gut health and systemic inflammation [111].
Quantitative Data on PEG400 Effects in Mice:
| Parameter Assessed | Control Group Findings | PEG400 Group Findings | Measurement Method |
|---|---|---|---|
| General Health | Lively, smooth fur, normal bowel movements | Quiet, weak, disheveled hair, persistent diarrhea, perianal redness/swelling | Clinical observation [111] |
| Body Weight | Stable | Significant decrease | Gravimetric measurement [111] |
| Colon Morphology | Compact mucosa, thick intestinal wall, tight folds | Wider colon cavity, thinner intestinal wall, relaxed folds, mucosal damage | Histopathology (H&E staining) [111] |
| Mucin Content (Muc2) | Normal level | Significant decrease | ELISA [111] |
| Inflammatory Markers | Normal IL-1β, normal IL-10 | Increased IL-1β (pro-inflammatory), decreased IL-10 (anti-inflammatory) | ELISA [111] |
| Gut Microbiota Alpha Diversity | Normal Chao1, Shannon, Simpson indices | Significantly decreased indices | 16S rRNA sequencing [111] |
| Firmicutes/Bacteroidetes Ratio | Normal ratio | 4.5x increase in ratio | 16S rRNA sequencing [111] |
| A. muciniphila Abundance | < 0.1% relative abundance | Increased to nearly 20% relative abundance | 16S rRNA sequencing [111] |
The experimental workflow and key causal relationships derived from such studies can be visualized as follows:
Underlying Mechanisms of Toxicity
The path from excipient exposure to systemic toxicity involves several interconnected biological pathways. The diagram below outlines the primary mechanistic relationship between the observed physiological changes.
Experimental Protocols for Safety Assessment
A multi-faceted approach is essential for comprehensively evaluating excipient safety, particularly concerning gut health and mineral absorption.
In Vivo Assessment of Intestinal Integrity and Inflammation
This protocol is designed to quantify changes in mucosal structure and inflammatory status in a rodent model [111].
- Animal Model: Use healthy adult mice (e.g., C57BL/6J). Divide into control (vehicle) and experimental (excipient) groups (n ≥ 6).
- Dosing Regimen: Administer the excipient via oral gavage daily for a defined period (e.g., 14 days). The dose should be relevant to proposed human exposure.
- Tissue Collection: Euthanize animals at endpoint. Immediately collect colon and intestinal segments.
- Histopathological Analysis:
- Fix intestinal segments in 10% neutral buffered formalin.
- Process, embed in paraffin, and section at 4-5 μm thickness.
- Stain with Hematoxylin and Eosin (H&E).
- Examine via light microscopy for structural integrity, crypt architecture, goblet cell population, and inflammatory cell infiltration. Score damage semi-quantitatively.
- Mucin and Cytokine Quantification:
- Homogenize snap-frozen intestinal tissue in appropriate buffer.
- Quantify Mucin 2 (Muc2) protein, a key component of the intestinal mucus layer, and pro/anti-inflammatory cytokines (e.g., IL-1β, IL-10) using commercially available Enzyme-Linked Immunosorbent Assay (ELISA) kits according to manufacturer protocols.
Gut Microbiota Analysis via 16S rRNA Sequencing
This protocol assesses the impact of an excipient on the composition and diversity of the gut microbiome [111].
- Sample Collection: Collect fecal pellets from experimental animals at baseline and endpoint. Snap-freeze in liquid nitrogen and store at -80°C.
- DNA Extraction: Isolate total genomic DNA from fecal samples using a dedicated stool DNA extraction kit.
- 16S rRNA Gene Amplification: Amplify the hypervariable regions (e.g., V3-V4) of the bacterial 16S rRNA gene using barcoded universal primers.
- Sequencing: Perform high-throughput sequencing on an Illumina MiSeq or HiSeq platform to generate paired-end reads.
- Bioinformatic Analysis:
- Process raw sequences (quality filtering, denoising, merging) using QIIME2 or Mothur.
- Cluster sequences into Operational Taxonomic Units (OTUs) or Amplicon Sequence Variants (ASVs) at a 97% similarity threshold.
- Assign taxonomy using a reference database (e.g., SILVA, Greengenes).
- Calculate alpha diversity indices (Chao1, Shannon, Simpson) and beta diversity (PCoA) to evaluate within-sample and between-sample diversity.
Metabolomic Profiling
This protocol identifies global changes in metabolic pathways induced by excipient exposure [111].
- Sample Preparation: Prepare serum or fecal extracts. For LC-MS, protein precipitation with cold acetonitrile is typical.
- Data Acquisition: Analyze samples using Liquid Chromatography coupled with Tandem Mass Spectrometry (LC-MS/MS) on a high-resolution instrument (e.g., Q-Exactive Orbitrap).
- Use both reverse-phase and HILIC chromatography to capture a broad range of metabolites.
- Acquire data in both positive and negative ionization modes.
- Data Processing and Analysis:
- Process raw data for peak picking, alignment, and normalization using software like XCMS or Progenesis QI.
- Perform multivariate statistical analysis, including Principal Component Analysis (PCA) and Orthogonal Projections to Latent Structures-Discriminant Analysis (OPLS-DA), to identify differentially abundant metabolites.
- Identify significant metabolites by searching against databases (e.g., HMDB, METLIN).
- Conduct pathway enrichment analysis (KEGG, MetaboAnalyst) to identify disrupted biological pathways (e.g., lipid and energy metabolism).
The following workflow summarizes the key stages of a comprehensive safety assessment:
Regulatory and Industrial Perspectives on Excipient Approval
Navigating the regulatory landscape is critical for the successful development of formulations containing novel excipients or those used in new contexts.
Regulatory Pathways and Strategic Considerations
Table: Pharmaceutical Excipient Development Pathways and Strategies
| Pathway / Aspect | Key Objective | Strategic Benefit | Risk Mitigation |
|---|---|---|---|
| 505(b)(2) / Hybrid Procedure | Leverage existing safety data for approved drugs for reformulations [112]. | Reduces risk, cost, and streamlines path to market for new delivery routes or chronic disease treatments [112]. | Relies on existing API data; focus shifts to demonstrating excipient safety and compatibility. |
| Novel Excipient Development | Achieve challenging technical targets (e.g., solubility, bioavailability) not possible with traditional excipients [112]. | Provides intellectual property protection and enables differentiated products [112]. | Requires comprehensive safety testing; collaboration with expert manufacturers is crucial. |
| Impurity Control (ICH M7, Q3B(R2)) | Determine safe limits of mutagenic compounds and impurities in pharmaceutical products [112]. | Provides a globally recognized framework for quality and safety, minimizing adverse effect potential [112]. | Requires rigorous supplier qualification and analytical testing protocols throughout the supply chain. |
| Functional Excipients (e.g., Lipids) | Improve bioavailability of low-solubility (BCS Class II/IV) APIs via systems like SEDDS [112]. | Enhances drug efficacy; enables dosage form transition to more economical and stable tablets [112]. | Requires careful formulation screening to optimize solubility and emulsion characteristics. |
The Scientist's Toolkit: Key Research Reagents and Materials
Table: Essential Materials for Excipient Safety and Bioavailability Research
| Reagent / Material | Function in Research |
|---|---|
| ELISA Kits (e.g., Muc2, IL-1β, IL-10) | Quantify specific proteins related to mucosal integrity and inflammatory response in tissue homogenates or serum [111]. |
| 16S rRNA Primers & Stool DNA Kit | Amplify and extract bacterial genomic DNA from fecal samples for subsequent microbiome sequencing analysis [111]. |
| LC-MS/MS Grade Solvents | Ensure high sensitivity and low background noise during metabolomic profiling of serum, feces, or tissue extracts [111]. |
| Functional Lipids (e.g., MCTs, Mono-/Diglycerides) | Serve as components of Self-Emulsifying Drug Delivery Systems (SEDDS) to study enhanced solubility and bioavailability of poorly soluble actives [112]. |
| Co-processed Excipient Systems (e.g., ABISORB-DC) | Enable the direct compression of lipid-based formulations into tablets, facilitating the study of stable and economical solid dosage forms [112]. |
| Formulation Screening Kits (e.g., ABISOL Kit) | Provide pre-formulated emulsion preconcentrates for high-throughput screening of API solubility and emulsion characteristics to fast-track formulation development [112]. |
| Histology Reagents (Formalin, H&E Stain) | Preserve and stain intestinal tissue sections for microscopic evaluation of mucosal structure and pathology [111]. |
The strategic development process for a formulation incorporating a novel excipient, from conception to regulatory submission, can be summarized as follows:
The integration of rigorous safety assessments and strategic regulatory planning is paramount in the development of excipients for mineral absorption research and pharmaceutical application. Empirical evidence demonstrates that excipients once presumed inert, such as PEG400, can exert significant detrimental effects on gut microbiota, mucosal integrity, and systemic metabolic homeostasis. These findings underscore the necessity of a comprehensive safety evaluation framework that includes gut microbiome analysis, metabolomic profiling, and detailed histopathological examination. Furthermore, the successful navigation of regulatory pathways and the application of novel, functional excipients offer a promising route to enhancing mineral bioavailability while ensuring patient safety. For researchers and drug development professionals, a deep understanding of these considerations is not merely a regulatory hurdle but a fundamental component of designing effective, safe, and innovative therapeutic solutions.
This whitepaper explores insulin, heparins, and bisphosphonates as paradigmatic models in drug development, framed within the context of inhibitors and enhancers of mineral absorption and skeletal homeostasis. These compounds, developed for metabolic and skeletal disorders, exhibit sophisticated mechanisms that directly and indirectly influence mineral ion metabolism, bone remodeling, and calcium-phosphate homeostasis. We provide a comprehensive technical analysis of their molecular mechanisms, experimental methodologies for evaluating their efficacy on mineralized tissues, and visualization of key signaling pathways. The data presented herein aim to offer researchers and drug development professionals a foundational framework for understanding how targeted therapies can modulate complex physiological processes involving mineral metabolism.
The development of therapeutic agents that influence mineral absorption and skeletal integrity represents a cornerstone of pharmaceutical science. Insulin, heparins, and bisphosphonates, though developed for distinct clinical indications (diabetes, anticoagulation, and osteoporosis, respectively), serve as exemplary models for understanding how molecules can be engineered to target specific pathways in mineral metabolism and bone remodeling. Osteoporosis, characterized by low bone mass and deterioration of bone structure, underscores the critical importance of bone homeostasis [113]. The bone remodeling unit, or basic multicellular unit (BMU), is the fundamental site where coupled resorption and formation occur, a process tightly regulated by systemic and local factors [114]. Disruption of this balance, as seen in drug-induced osteoporosis or metabolic diseases, can lead to significant fragility fractures [113] [115]. This review delves into the mechanistic insights, experimental protocols, and developmental trajectories of these three model compounds, highlighting their role as both enhancers and inhibitors within the mineral absorption research paradigm.
Bone Remodeling and Mineral Homeostasis: A Primer
Bone remodeling is a continuous, lifelong process essential for maintaining skeletal integrity, repairing micro-damage, and regulating systemic mineral ion homeostasis, particularly calcium and phosphate [114] [116]. This cycle involves sequential phases of activation, resorption, reversal, formation, and mineralization, terminated by a return to quiescence [116].
The process is orchestrated by a complex interplay of bone cells:
- Osteoclasts: Multinucleated cells of hematopoietic origin responsible for bone resorption. They secrete enzymes like tartrate-resistant acid phosphatase (TRAP) and cathepsin K to degrade the mineralized bone matrix [116].
- Osteoblasts: Mesenchymal lineage-derived cells responsible for bone formation. They secrete collagenous and non-collagenous proteins to form the osteoid, which subsequently undergoes mineralization [114] [116]. Their differentiation is regulated by key transcription factors like Runx2 [116].
- Osteocytes: Terminally differentiated osteoblasts entombed within the bone matrix. They act as mechanosensors and endocrine cells, secreting factors like sclerostin (a Wnt pathway inhibitor) and RANKL to regulate both osteoblast and osteoclast activity [114] [116].
Critical to the regulation of this process are several key signaling pathways, including the RANK/RANKL/OPG pathway controlling osteoclastogenesis, and the Wnt/β-catenin pathway promoting osteoblastogenesis and bone formation [116]. The following diagram illustrates the core regulatory and cellular framework of bone remodeling.
Diagram 1: Core regulatory network in bone remodeling. Osteocytes respond to stimuli by secreting factors that control osteoclast and osteoblast activity. The RANKL/RANK/OPG axis is a primary driver of osteoclastogenesis, while the Wnt/β-catenin pathway is crucial for bone formation. PTH = Parathyroid Hormone.
Case Study 1: Bisphosphonates
Background and Clinical Indication
Bisphosphonates are first-line anti-resorptive agents for treating osteoporosis and other skeletal disorders characterized by excessive bone loss, such as those induced by glucocorticoids [114] [115]. They are also effective in managing bone metastases. Their development shifted the osteoporosis treatment paradigm by offering potent inhibition of bone loss and significant fracture risk reduction [114].
Mechanism of Action: Molecular Targets and Mineral Effects
Bisphosphonates are synthetic analogs of inorganic pyrophosphate (P-C-P backbone instead of P-O-P) with high affinity for bone mineral surfaces, particularly at active resorption sites [117]. Their primary action is the direct inhibition of osteoclast-mediated bone resorption. The molecular mechanism depends on the specific side chain (R2 group):
- Nitrogen-containing bisphosphonates (e.g., Alendronate, Risedronate, Zoledronate): These are the most potent. They inhibit the mevalonate pathway by targeting farnesyl diphosphate synthase (FPP synthase). This prevents the prenylation of small GTPases (e.g., Ras, Rho, Rac), which are essential for osteoclast cytoskeletal organization, membrane ruffling, and survival, leading to osteoclast apoptosis [117].
- Non-nitrogen-containing bisphosphonates (e.g., Etidronate, Clodronate): These are metabolically incorporated into non-hydrolysable analogues of ATP (AppCp-type molecules). The accumulation of these cytotoxic metabolites within osteoclasts induces apoptosis [117].
By potently inhibiting osteoclasts, bisphosphonates reduce the release of calcium and phosphate from the bone matrix into the circulation, effectively acting as inhibitors of mineral mobilization from the skeletal reservoir.
Key Experimental Protocols
Protocol for Evaluating In Vitro Osteoclast Activity:
- Cell Culture: Differentiate osteoclasts from murine RAW 264.7 cell line or human peripheral blood mononuclear cells (PBMCs) using 50 ng/mL RANKL and 25 ng/mL M-CSF for 5-7 days.
- Treatment: Add varying concentrations of the test bisphosphonate (e.g., 1 µM–100 µM) to the culture medium during differentiation.
- Resorption Assay: Seed cells on calcium phosphate-coated plates (e.g., Corning Osteo Assay Surface). After treatment, remove cells with 5% sodium hypochlorite.
- Analysis: Quantify resorption pits by imaging with light microscopy or scanning electron microscopy. Analyze the percentage of resorbed area using image analysis software (e.g., ImageJ).
- Viability/Apoptosis Assay: Perform parallel assays using MTT or Alamar Blue for cell viability and Annexin V/propidium iodide staining for apoptosis via flow cytometry.
Protocol for In Vivo Efficacy in Rodent Models of Osteoporosis:
- Animal Model: Use 3-month-old female Sprague-Dawley rats subjected to ovariectomy (OVX) to induce estrogen-deficient osteoporosis. Sham-operated rats serve as controls.
- Dosing: Administer bisphosphonate (e.g., Alendronate, 5–50 µg/kg) or vehicle subcutaneously 2-3 times per week, starting one week post-surgery, for 12 weeks.
- Sample Collection: At endpoint, collect blood serum for bone turnover markers (e.g., CTX-1 for resorption, P1NP for formation) and dissect femurs and tibiae.
- Micro-Computed Tomography (µCT): Scan excised femurs at an isotropic resolution of 10-20 µm. Analyze trabecular bone parameters in the distal femoral metaphysis, including Bone Volume/Total Volume (BV/TV), Trabecular Number (Tb.N), and Trabecular Separation (Tb.Sp).
- Histomorphometry: Embed undecalcified bone in methyl methacrylate. Section and stain with Toluidine Blue or Goldner's Trichrome for static parameters (e.g., osteoclast surface/bone surface, Oc.S/BS) and with calcein double-labeling for dynamic parameters of bone formation (e.g., mineral apposition rate, MAR).
Quantitative Efficacy Data
Table 1: Quantitative effects of selected bisphosphonates on Bone Mineral Density (BMD) and fracture risk in clinical trials.
| Drug (Example) | Dosage Regimen | Increase in Lumbar Spine BMD (%) | Fracture Risk Reduction (Non-vertebral) | References |
|---|---|---|---|---|
| Alendronate | 70 mg once weekly | ~4%/year | 20–30% | [114] |
| Risedronate | 35 mg once weekly | ~3%/year | 20–30% | [114] |
| Ibandronate | 150 mg once monthly | ~3%/year | Not significant for non-vertebral | [114] |
| Zoledronic Acid | 5 mg IV/year | ~4%/year | 20–30% | [114] |
Research Reagent Solutions
Table 2: Key reagents for studying bisphosphonate mechanisms and efficacy.
| Research Reagent | Function/Application | Supplier Examples |
|---|---|---|
| RAW 264.7 Cell Line | Murine macrophage cell line used as a model for in vitro osteoclast differentiation. | ATCC |
| Recombinant RANKL | Cytokine essential for inducing osteoclast differentiation and activation in cell cultures. | R&D Systems, PeproTech |
| Recombinant M-CSF | Cytokine required for the survival and proliferation of osteoclast precursors. | R&D Systems, PeproTech |
| Corning Osteo Assay Surface | 96-well plates coated with a synthetic calcium phosphate film for quantifying osteoclast resorptive activity. | Corning Inc. |
| Alendronate Sodium | A potent nitrogen-containing bisphosphonate used as a reference standard in experimental studies. | Sigma-Aldrich, Tocris |
| Farnesyl Diphosphate Synthase Assay Kit | For directly measuring the enzymatic activity of FPP synthase, the key molecular target of N-BPs. | Abcam |
Case Study 2: Heparins
Background and Clinical Indication
Unfractionated heparin (UFH) is a rapid-acting parenteral anticoagulant used for the treatment and prevention of thromboembolic diseases. A significant adverse effect of long-term (≥6 months), high-dose (>15,000 units/day) UFH therapy is drug-induced osteoporosis [113] [115]. This side effect profile makes heparins a critical case study of a therapeutic agent that inadvertently acts as a potent inhibitor of bone integrity.
Mechanism of Action: Molecular Targets and Mineral Effects
The precise cellular mechanism of heparin-induced bone loss is not fully elucidated but involves several pathways that disrupt the bone remodeling balance:
- Increased Bone Resorption: Heparin stimulates osteoclast formation and activity, potentially by enhancing RANKL signaling or other pro-osteoclastogenic pathways [113].
- Decreased Bone Formation: Heparin suppresses osteoblast function and reduces osteoblast differentiation, leading to diminished bone formation [113].
- Other Proposed Mechanisms: These include depletion of bone marrow mast cells and enhancement of parathyroid hormone (PTH) function, which can promote calcium release from bone [113].
Notably, low-molecular-weight heparins (LMWHs), such as enoxaparin, appear to have a significantly lower risk of inducing osteoporosis compared to UFH, making them a safer alternative for long-term anticoagulation [113].
Key Experimental Protocols
Protocol for Assessing Heparin-Induced Osteoclastogenesis:
- Co-culture System: Establish a co-culture of murine bone marrow cells (as osteoclast precursors) and primary osteoblasts (which provide necessary RANKL and M-CSF).
- Treatment: Treat co-cultures with varying concentrations of UFH (e.g., 1–100 µg/mL) and a positive control (e.g., RANKL+M-CSF). Include an LMWH for comparison.
- Staining and Quantification: After 7-10 days, fix cells and perform staining for Tartrate-Resistant Acid Phosphatase (TRAP). Count the number of multinucleated (≥3 nuclei) TRAP-positive cells as mature osteoclasts.
Protocol for In Vivo Assessment of Heparin on Bone Mass:
- Animal Model: Use 8-week-old female C57BL/6 mice or Sprague-Dawley rats.
- Dosing: Administer UFH (e.g., 1–2 U/g body weight) or LMWH subcutaneously once or twice daily for 4-6 weeks. A vehicle-treated group serves as control.
- Analysis: At termination, analyze trabecular bone architecture of the lumbar vertebrae (L3-L5) and distal femur via µCT to quantify BV/TV, Tb.N, and Tb.Sp.
- Serum Biomarkers: Measure serum levels of C-telopeptide of type I collagen (CTX-1) as a marker of bone resorption and osteocalcin for bone formation via ELISA.
Case Study 3: Insulin
Background and Clinical Indication
Insulin is a peptide hormone essential for the treatment of type 1 and advanced type 2 diabetes. Its role in bone and mineral metabolism is complex and multifactorial. While insulin itself is not a direct therapy for bone diseases, insulin resistance in type 2 diabetes is a significant risk factor for skeletal fragility and increased fracture risk, despite often normal or high BMD [118]. This paradox highlights the intricate connection between energy metabolism and bone homeostasis.
Mechanism of Action: Molecular Targets and Mineral Effects
Insulin influences bone through direct and indirect mechanisms:
- Direct Anabolic Effects: Osteoblasts express insulin receptors. Insulin signaling in these cells promotes glucose uptake, collagen synthesis, and overall osteoblast activity and differentiation, thereby enhancing bone formation and mineralization [118].
- Indirect Effects via the IGF-1 Axis: Insulin stimulates the production of Insulin-like Growth Factor-1 (IGF-1) in the liver and bone. IGF-1 is a potent anabolic agent for bone, stimulating osteoblast proliferation and function.
- Coupling to Energy Metabolism: The skeleton is now recognized as an endocrine organ. Osteocalcin, one of the most abundant non-collagenous proteins in bone, undergoes carboxylation in a vitamin K-dependent process. Insulin signaling in osteoblasts can decarboxylate osteocalcin, activating it as a hormone that promotes pancreatic beta-cell insulin secretion and improves glucose metabolism, creating a bone-pancreas feedback loop.
Therefore, insulin deficiency or resistance disrupts this integrated network, leading to impaired bone formation and potentially altered mineral homeostasis.
Key Experimental Protocols
Protocol for Investigating Insulin Signaling in Osteoblasts:
- Cell Culture: Use a human osteoblast cell line (e.g., SaOS-2, hFOB 1.19) or primary human osteoblasts.
- Serum Starvation: Starve cells in serum-free medium for 6-12 hours to synchronize cell cycles and quiesce signaling pathways.
- Stimulation: Stimulate cells with 100 nM insulin for 5, 15, 30, and 60 minutes. Include an insulin receptor inhibitor (e.g., HNMPA) as a control.
- Western Blot Analysis: Lyse cells and perform Western blotting to detect phosphorylation states of key signaling molecules in the insulin receptor pathway, including the Insulin Receptor β-subunit (Tyr1150/1151), IRS-1, AKT (Ser473), and ERK1/2 (Thr202/Tyr204).
- Functional Assays: Assess downstream functional outcomes, such as alkaline phosphatase (ALP) activity, osteocalcin mRNA expression (via qPCR), or mineralized nodule formation (via Alizarin Red S staining), after several days of insulin treatment.
Protocol for Assessing Bone Phenotype in Diabetic Rodent Models:
- Animal Model: Use leptin receptor-deficient mice (db/db mice, a model of type 2 diabetes) or streptozotocin (STZ)-induced diabetic rats (a model of type 1 diabetes).
- Intervention: Treat diabetic animals with insulin via subcutaneous implants (slow-release) or daily injection to achieve euglycemia for 8-12 weeks. Compare to untreated diabetic and non-diabetic wild-type controls.
- Analysis:
- µCT: Analyze trabecular and cortical bone architecture in the femur and lumbar spine.
- Biomechanical Testing: Perform 3-point bending tests on the femoral diaphysis to assess bone strength (ultimate load, stiffness).
- Histomorphometry: Perform dynamic bone histomorphometry with calcein double labeling to measure bone formation rates (BFR/BS).
The following diagram illustrates the key signaling pathways discussed in these case studies, highlighting their points of convergence and divergence in regulating bone cell activity.
Diagram 2: Molecular targets and cellular outcomes for insulin, heparins, and bisphosphonates. The diagram contrasts the anabolic insulin signaling in osteoblasts with the anti-resorptive action of bisphosphonates in osteoclasts and the uncoupling effect of heparin, which promotes resorption and inhibits formation. N-BPs = Nitrogen-containing Bisphosphonates; FPP = Farnesyl Diphosphate; IGF-1 = Insulin-like Growth Factor 1.
Discussion and Future Perspectives
The comparative analysis of insulin, heparins, and bisphosphonates reveals a sophisticated interplay between pharmacology and mineral metabolism. Bisphosphonates exemplify a targeted inhibitor of osteoclast-mediated mineral release, designed specifically to enhance bone mass by suppressing resorption. In contrast, long-term heparin therapy serves as a cautionary tale of an unintended inhibitor of skeletal integrity, highlighting the importance of monitoring bone health during drug development for non-skeletal indications. Insulin represents a more nuanced enhancer of bone formation through direct and systemic anabolic signaling, though its benefits are contingent on proper metabolic control.
Future therapeutic strategies are increasingly focusing on anabolic agents that enhance bone formation beyond what is possible with anti-resorptives. Drugs like teriparatide (recombinant PTH 1-34) represent this class, stimulating osteoblast activity more effectively than bisphosphonates in high-risk patients, including those with glucocorticoid-induced osteoporosis [114] [115]. Furthermore, novel drug candidates like SR10171, a next-generation PPARγ modulator, demonstrate the potential for a single agent to treat both type 2 diabetes and osteoporosis by improving bone mass and insulin sensitivity concurrently [118]. This "targeted polypharmacology" approach, which aims to engage molecular targets in unique ways to achieve multiple therapeutic benefits, is a promising frontier in drug development for metabolic bone diseases and beyond.
The development of mineral-based therapeutics and supplements represents a critical field at the intersection of nutrition and pharmaceutical science. Achieving economic and practical viability for these products requires successfully navigating three interconnected challenges: manufacturing scalability, product stability, and patient compliance. Within the broader context of mineral absorption research, understanding the inhibitors and enhancers of bioavailability is fundamental to formulating effective products that maintain their therapeutic integrity from production through patient consumption. This technical guide examines these critical factors, providing researchers and drug development professionals with evidence-based methodologies and practical frameworks to optimize mineral-based product development.
The efficacy of mineral supplements depends not only on the total mineral content but also on the fraction absorbed and utilized by the body [65]. This bioavailability is influenced by multiple factors throughout the product lifecycle, from the chemical form of the mineral and its formulation composition to its stability profile and eventual patient adherence to dosing regimens. A holistic approach that integrates absorption science with robust manufacturing and stability practices is essential for developing viable mineral-based health products.
Manufacturing Scalability for Mineral Products
Scale-Up Challenges and Formulation Considerations
The transition from laboratory-scale formulation to commercial manufacturing presents significant technical challenges that can impact both the economic viability and absorption characteristics of mineral products. Research efforts in early development frequently fail to consider the complexities of scaling production, creating major hurdles later in the drug development process [119]. The primary objectives during scale-up include maintaining consistent product quality, ensuring content uniformity, and preserving the dissolution properties that affect mineral bioavailability.
Common scalability challenges include:
- Flowability Issues: Powder blends with poor flow characteristics compromise content uniformity in tablets and capsules. This is particularly problematic with high-dose minerals and micronized active pharmaceutical ingredients (APIs) that exhibit cohesive properties [120].
- Cohesivity and Static Charges: Hydrophobic, micronized APIs often develop static charges and adhere to processing equipment surfaces, leading to material loss and content uniformity issues [120].
- Process-Induced Transformations: Mechanical stresses during granulation, milling, and compression can potentially alter the physical form of minerals, potentially affecting their dissolution and absorption characteristics.
Practical Engineering and Formulation Solutions
Addressing scale-up challenges requires both formulation optimization and engineering interventions. The table below summarizes key challenges and validated solutions:
Table 1: Scale-Up Challenges and Solutions for Mineral Product Manufacturing
| Challenge | Formulation Solutions | Engineering/Process Solutions |
|---|---|---|
| Poor Powder Flow | Transition to granulation (dry/wet/fluid bed); Incorporate flow aids (colloidal silicon dioxide); Use co-processed excipients [120] | Double transition hopper design; Vibratory mechanisms; Paddle stirrers in hoppers [120] |
| Content Uniformity | Optimize binder selection (HPC, HPMC, PVP); Use appropriate lubricants/glidants (Mg/Ca stearate) [120] | Mass flow optimization; Forced feeder systems on tablet presses; Optimized impeller geometry [120] |
| Sticking/Adhesion | Modify lubricant type/particle size; Incorporate anti-adherents; Functional coating systems [120] | Tooling coatings (CrN, TiN); Process parameter modulation (compression force, dwell time); Equipment grounding [120] |
For mineral products specifically, the selection of mineral salts (e.g., citrates, gluconates, bisglycinates) versus traditional inorganic forms (e.g., oxides, carbonates) can significantly impact both absorption potential and processing characteristics. While chelated minerals typically demonstrate enhanced absorption due to protection from dietary inhibitors, their flow and compaction properties may differ substantially from conventional forms, necessitating tailored formulation approaches.
Stability Studies for Mineral-Based Products
Stability Testing Fundamentals
Stability studies are essential for determining shelf life, appropriate storage conditions, and packaging requirements for mineral products, ensuring they remain safe, effective, and within specified quality attributes throughout their shelf life [121]. These studies evaluate how environmental factors like temperature, humidity, and light impact product quality over time [121]. The International Council for Harmonisation (ICH) provides comprehensive guidelines governing stability testing through several key documents, with ICH Q1A(R2) outlining core stability testing requirements [121].
The pharmaceutical industry recognizes three primary types of stability studies:
- Accelerated Studies: Expose products to elevated temperatures (typically 40°C and 75% relative humidity) for up to six months to predict long-term stability and identify degradation mechanisms [121].
- Intermediate Studies: Conducted at 30°C and 65% RH for six months when accelerated studies indicate significant changes, helping refine long-term predictions [121].
- Long-term Studies: Evaluate product stability under typical storage conditions, often 25°C and 60% RH for a minimum of 12 months, though duration may extend based on proposed shelf life [121].
Key Parameters and Methodologies in Stability Testing
Stability testing for mineral products encompasses multiple quality attributes:
Table 2: Key Stability Testing Parameters for Mineral-Based Products
| Testing Category | Specific Parameters Assessed | Analytical Methodologies |
|---|---|---|
| Physical Characteristics | Appearance, color, clarity, physical state, hardness, friability, dissolution rates [121] | Visual inspection, dissolution apparatus, texture analysis, disintegration testing |
| Chemical Attributes | API content, degradation products, impurity profiles, potency [121] | HPLC, mass spectrometry, spectrophotometry, titration |
| Microbiological Properties | Microbial limits, sterility, preservative effectiveness, bioburden [121] | Microbial enumeration tests, sterility testing, antimicrobial effectiveness testing |
| Container Closure Evaluation | Material compatibility, moisture permeation, light protection, extractables/leachables [121] [122] | Extraction studies, leachable studies, moisture permeation testing |
Stability testing protocols must include validated analytical methods that demonstrate specificity, accuracy, precision, linearity, and robustness for stability samples [121]. Testing frequency follows a predetermined schedule, with more frequent testing during early time points and extended intervals as the study progresses [121].
Factors Influencing Product Stability
Multiple factors determine the stability profile of mineral-based products:
- Storage Conditions: Temperature, relative humidity, and light exposure significantly impact stability. Higher temperatures and humidity accelerate deterioration, while light exposure can increase decomposition, particularly for photosensitive compounds [123].
- Dosage Form: Liquid dosage forms containing water are more prone to degradation compared to solid dosage forms due to increased susceptibility to hydrolysis and microbial growth [123].
- Container Closure System: The moisture-barrier properties of primary packaging critically influence stability by controlling humidity exposure [123]. Semi-permeable containers require evaluation of potential water loss in addition to other stability parameters [123].
- Storage Time: As a general rule, medicinal products tend to degrade over time, with longer storage increasing the likelihood of degradation [123].
Stability Influence Factors
Patient Compliance in Mineral Supplementation
Adherence Assessment and Challenges
Patient compliance with prescribed mineral supplementation regimens represents a critical factor in determining therapeutic success, particularly in chronic conditions requiring long-term use. A two-year cohort study of bariatric surgery patients, who require lifelong vitamin and mineral supplementation, revealed substantial variation in adherence patterns [124] [125]. While approximately 60% of study participants maintained high adherence rates (≥80%) two years post-surgery, a significant proportion discontinued their intended lifelong supplementation: 15% for calcium-vitamin D and 12% for vitamin B12 [124] [125].
Several factors associated with low adherence have been identified:
- Younger age
- Experience of side effects
- Mental health problems
- No regular use of medication before surgery [124] [125]
These findings highlight the need for tailored follow-up and strategies to support long-term persistence with mineral supplementation regimens [124] [125].
Formulation Strategies to Enhance Compliance
Several formulation approaches can improve patient adherence to mineral supplementation:
- Dosage Form Optimization: Patient-friendly dosage forms such as once-daily tablets, chewable formulations, or liquid concentrates can significantly improve adherence, particularly for populations with difficulty swallowing conventional solid dosage forms.
- Reduced Pill Burden: Combination formulations that incorporate multiple minerals in a single dosage unit can decrease the number of supplements patients need to take daily.
- Side Effect Mitigation: Formulation approaches that minimize gastrointestinal irritation, such as enteric coating or chelated mineral forms, can improve tolerability and long-term adherence.
- Patient-Centric Packaging: Compliance-enhancing packaging with clear administration instructions, calendar blister packs, and reminder systems can support consistent supplementation.
Advanced Considerations in Mineral Absorption
Mineral Absorption Mechanisms
The bioavailability of minerals depends on their absorption mechanisms in the gastrointestinal tract. Mineral absorption occurs through two primary pathways:
- Paracellular Pathway: Passive diffusion between intestinal epithelial cells, driven by concentration gradients [3].
- Transcellular Pathway: Active transport across intestinal epithelial cells via specific transporters and channels [3].
Specific transporters facilitate mineral uptake, such as TRPV6 for calcium and ZIP4 for zinc [3]. Some minerals may undergo exchange with other ions during absorption; for example, zinc absorption can be influenced by exchange with other divalent cations like copper [3]. Understanding these mechanisms is essential for formulating minerals in ways that enhance their absorption and bioavailability.
Probiotics as Mineral Absorption Enhancers
Emerging research indicates that probiotics can significantly modulate mineral absorption through several mechanisms:
- Phytate Degradation: Certain probiotic strains produce phytase enzymes that break down phytic acid, a compound in plant-based foods that binds minerals and inhibits their absorption [3]. Phytase-producing microorganisms include Bacillus subtilis, Bacillus coagulans, and various Lactobacillus species (acidophilus, fermentum, plantarum) [3].
- Mineral Solubilization: Probiotics can produce short-chain fatty acids and other metabolites that acidify the intestinal environment, potentially increasing mineral solubility and absorption [3].
- Intestinal Barrier Function: Some evidence suggests that certain probiotics may enhance mineral absorption by improving intestinal barrier function and increasing the expression of mineral transporters [3].
The effects of probiotics on mineral absorption are strain-specific and depend on factors including dosage, duration of supplementation, and individual differences in gut microbiota composition [3]. This represents a promising area for developing mineral formulations with enhanced bioavailability.
Research Reagent Solutions for Mineral Absorption Studies
Table 3: Essential Research Reagents for Mineral Absorption Studies
| Reagent/Category | Specific Examples | Research Application |
|---|---|---|
| Mineral Standards | Calcium citrate, Zinc bisglycinate, Magnesium oxide, Ferrous sulfate | Reference compounds for bioavailability comparisons and analytical method development |
| Probiotic Strains | Lactobacillus acidophilus, Bifidobacterium bifidum, Bacillus subtilis | Investigation of mineral absorption enhancement mechanisms [3] |
| Transport Inhibitors | Ruthenium red (TRPV6 inhibitor), ZIP4 blockers | Mechanistic studies to elucidate specific absorption pathways [3] |
| Analytical Standards | Stable isotope tracers (⁶⁷Zn, ⁴⁴Ca), Certified reference materials | Precise quantification of mineral absorption and distribution |
| Cell Culture Models | Caco-2 cell lines, HT-29-MTX co-cultures | In vitro assessment of mineral transport and absorption mechanisms |
Integrated Development Framework
Successful development of mineral-based products requires an integrated approach that connects absorption science with practical development considerations. The following diagram illustrates the interconnected factors influencing mineral product viability:
Mineral Product Development Framework
This framework demonstrates how absorption science should inform formulation development, which subsequently impacts manufacturing approach and stability profile, ultimately influencing patient compliance and therapeutic efficacy. A disruption at any point in this continuum compromises the overall viability of the mineral product.
The development of economically and practically viable mineral-based products requires a multidisciplinary approach that integrates absorption science, formulation technology, manufacturing engineering, stability assessment, and patient behavior understanding. Successful products must balance chemical stability with optimal bioavailability while maintaining manufacturability and supporting patient adherence.
Emerging research on mineral absorption enhancers, particularly probiotics, offers promising avenues for developing next-generation mineral formulations with superior bioavailability profiles. Simultaneously, advances in manufacturing technologies and stability testing methodologies continue to improve the efficiency and reliability of mineral product development.
By adopting the integrated framework and methodologies presented in this technical guide, researchers and drug development professionals can more effectively navigate the complex interplay of factors that determine the ultimate success of mineral-based therapeutics and supplements, thereby delivering products that demonstrate both scientific excellence and practical viability in real-world settings.
Conclusion
The intricate landscape of mineral absorption presents both significant challenges and remarkable opportunities for biomedical research and therapeutic development. A multidisciplinary approach that integrates foundational knowledge of absorption mechanisms with advanced enhancement technologies is essential for overcoming bioavailability barriers. The convergence of nutritional science, pharmaceutical technology, and microbiome engineering offers promising pathways for developing next-generation mineral delivery systems with improved efficacy and safety profiles. Future research should prioritize the validation of predictive bioavailability models, exploration of personalized nutrition approaches based on genetic and microbial biomarkers, and translation of laboratory innovations into clinically viable therapies. Addressing global mineral deficiencies and optimizing mineral-based therapeutics will require continued collaboration across disciplines to develop targeted solutions that account for individual variability and complex nutrient-drug interactions.
References
- 1. Invited review: Mineral absorption mechanisms ... [https://www.sciencedirect.com/science/article/pii/S0022030218300614]
- 2. Editorial: Bioactive compounds in mineral bioavailability [https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2022.1050670/full]
- 3. Probiotic-driven advancement: Exploring the intricacies of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10767166/]
- 4. Mechanisms and implications of the gut microbial ... [https://www.nature.com/articles/s44324-025-00066-1]
- 5. An Intestinal Paracellular Pathway Biased Toward ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6219475/]
- 6. Intestinal distribution of transcripts regulating calcium and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12275116/]
- 7. Impact of dietary components and processing on mineral ... [https://www.sciencedirect.com/science/article/abs/pii/S0963996925017855]
- 8. Is There Such a Thing as “Anti-Nutrients”? A Narrative Review ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7600777/]
- 9. Reduction of phytic acid and enhancement of bioavailable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4325021/]
- 10. Anti-Nutrients Explained: Oxalates, Lectins And 'Toxic' ... [https://www.forbes.com/sites/christopherthompson/2025/10/08/anti-nutrients-explained-oxalates-lectins-and-toxic-vegetables/]
- 11. Are Anti-Nutrients Harmful? - The Nutrition Source [https://nutritionsource.hsph.harvard.edu/anti-nutrients/]
- 12. Oxalate: effect on calcium absorbability [https://www.sciencedirect.com/science/article/pii/S0002916523436349]
- 13. Dependence of oxalate absorption on the daily calcium ... [https://pubmed.ncbi.nlm.nih.gov/15153567/]
- 14. The Impact of Tannin Consumption on Iron Bioavailability ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5998341/]
- 15. Are tannins a double-edged sword in biology and health? [https://www.sciencedirect.com/science/article/abs/pii/S0924224498000284]
- 16. Foods High in Oxalates [https://www.webmd.com/diet/foods-high-in-oxalates]
- 17. Anti-nutrients of plant-based food [https://www.sciencedirect.com/science/article/pii/S2772566925003088]
- 18. Micronutrient bioavailability: concepts, influencing factors, ... [https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2025.1646750/full]
- 19. Trace Mineral Imbalances in Global Health [https://www.mdpi.com/2072-6643/17/13/2241]
- 20. Iron Metabolism, Calcium, Magnesium and Trace Elements [https://link.springer.com/article/10.1007/s12011-024-04289-z]
- 21. Leafy vegetables as a significant source of minerals [https://www.sciencedirect.com/science/article/abs/pii/S0924224425002079]
- 22. Synergistic effects produced by certain antioxidants in ... [https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2025.1558597/full]
- 23. Review of processes for improving the bioaccessibility ... [https://www.sciencedirect.com/science/article/pii/S2772753X23003891]
- 24. Framework for Developing Prediction Equations ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12361767/]
- 25. How anti-nutrients are sabotaging mineral absorption [https://www.supplysidesj.com/natural-product-development/lost-in-digestion-how-anti-nutrients-are-sabotaging-mineral-article]
- 26. Impact of Polymorphisms in Genes Related to Vitamin D ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12423549/]
- 27. Examining the association between genetic ... [https://www.sciencedirect.com/science/article/pii/S2352914825000401]
- 28. Association analysis between nutritional factors within the ... [https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2025.1592974/full]
- 29. The impact of single nucleotide polymorphisms on the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12628764/]
- 30. A Comprehensive Review of the Triangular Relationship ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12469625/]
- 31. Resilience to Global Health Challenges Through ... [https://www.mdpi.com/2072-6643/17/3/396]
- 32. Host genetic factors related to innate immunity ... [https://www.nature.com/articles/s41467-022-33906-5]
- 33. Clinical assessment of acid-base status. Strong ion difference theory - PubMed [https://pubmed.ncbi.nlm.nih.gov/10573806/]
- 34. Clinical Assessment of Acid-Base Status: Strong Ion Difference Theory - ScienceDirect [https://www.sciencedirect.com/science/article/abs/pii/S0749072015301584]
- 35. Physiology, Acid Base Balance - StatPearls - NCBI Bookshelf [https://www.ncbi.nlm.nih.gov/books/NBK507807/]
- 36. Nutritional disturbance in acid–base balance and osteoporosis: a hypothesis that disregards the essential homeostatic role of the kidney - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3828631/]
- 37. Phytate and Mineral Absorption: An Updated Perspective [https://sniglobal.org/phytate-and-mineral-absorption-an-updated-perspective/]
- 38. Bioaccessibility mechanisms, fortification strategies ... [https://www.sciencedirect.com/science/article/pii/S266683352400131X]
- 39. Quantitative Analysis of Mixed Minerals with Finite Phase ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10096478/]
- 40. Tracing metabolic flux to assess optimal dietary protein and ... [https://www.nature.com/articles/s12276-022-00817-w]
- 41. Investigating the Digestibility, Bioavailability and Utilization ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12609865/]
- 42. Investigating the Digestibility, Bioavailability and Utilization ... [https://pubmed.ncbi.nlm.nih.gov/41228401/]
- 43. Bioavailability enhancers of herbal origin: An overview - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3634921/]
- 44. Absorption Enhancers: Applications and Advances - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3291189/]
- 45. Clinically Relevant Herb-Micronutrient Interactions [https://www.sciencedirect.com/science/article/pii/S2161831322012480]
- 46. In silico analyses as a tool for regulatory assessment of ... [https://ui.adsabs.harvard.edu/abs/2025CmTox..3500372R/abstract]
- 47. In silico analyses as a tool for regulatory assessment of ... [https://www.sciencedirect.com/science/article/abs/pii/S2468111325000325]
- 48. In Silico Modelling of Physiologically Based Intestinal ... [https://www.sciencedirect.com/science/article/abs/pii/S026087742500398X]
- 49. Study on Mechanism of Surfactant Adsorption at Oil–Water ... [https://www.mdpi.com/1420-3049/30/12/2541]
- 50. Mechanisms and Effects of Bitumen Liberation in the ... [https://www.sciencedirect.com/science/article/abs/pii/S0927775725026184]
- 51. Bio-Inspired Muco-Adhesive Polymers for Drug Delivery ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9785024/]
- 52. Mucoadhesive Polymer-Based Formulations for Drug ... [https://www.mathewsopenaccess.com/full-text/mucoadhesive-polymer-based-formulations-for-drug-delivery]
- 53. sciencedirect.com/topics/medicine-and-dentistry/ mineral - absorption [https://www.sciencedirect.com/topics/medicine-and-dentistry/mineral-absorption]
- 54. Protein-Derived Chelating Peptides [https://pubmed.ncbi.nlm.nih.gov/40928017/]
- 55. Exploring the potential of polymers: advancements in oral ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12522156/]
- 56. Development of iron–ascorbic acid microcapsules using ... [https://pubs.rsc.org/en/content/articlehtml/2025/fb/d5fb00121h]
- 57. Harnessing lipid based nanocochleates for targeted ... [https://www.sciencedirect.com/science/article/abs/pii/S0927776525007945]
- 58. Lipid-Based Drug Delivery Systems: Concepts and Recent ... [https://www.mdpi.com/2079-4991/15/17/1326]
- 59. Lipid-based nano-carriers for the delivery of anti-obesity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12057057/]
- 60. Encapsulation Techniques of Carotenoids and Their ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12121523/]
- 61. Acoustic technologies for the orchestration of cellular functions ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12273796/]
- 62. Enhancing cancer therapy via acoustics - RSC Publishing [https://pubs.rsc.org/en/content/articlehtml/2025/lc/d5lc00419e]
- 63. Impact of chemical structure, lipidation and formulation on ... [https://www.sciencedirect.com/science/article/abs/pii/S0168365925007552]
- 64. Impact of Traditional Food Processing Techniques on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12191477/]
- 65. Framework for Developing Prediction Equations ... [https://www.sciencedirect.com/science/article/pii/S2161831325001176]
- 66. Impact of hydrothermal processing on the mineral ... [https://link.springer.com/article/10.1007/s44403-025-00027-y]
- 67. Fermentation-driven bioactive enhancement in cereal grains [https://www.sciencedirect.com/science/article/abs/pii/S0924224425005394]
- 68. Frontiers | Complexation of multiple mineral elements by fermentation ... [https://www.frontiersin.org/journals/nutrition/articles/10.3389/fnut.2022.1001412/full]
- 69. Effect of fermentation of chosen vegetables on the nutrient ... [https://www.nature.com/articles/s41598-022-17782-z]
- 70. Nutritional power couples [https://www.health.harvard.edu/nutrition/nutritional-power-couples]
- 71. Nutrient Synergy: What To Take Together For Maximum ... [https://www.iherb.com/blog/nutritional-synergy/2208?srsltid=AfmBOop5NDUcdwXsDm5m7HD0TLJETjbQcK6euQIEyrPjvHJwjoQ5BmrR]
- 72. Insight into phytase-producing microorganisms for phytate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10128089/]
- 73. Enhancing micronutrient absorption through simultaneous ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10290024/]
- 74. Bioavailability of iron, zinc, and other trace minerals from ... [https://www.sciencedirect.com/science/article/pii/S0002916522033652]
- 75. Microbial phytase: Their sources, production, and role in ... [https://www.sciencedirect.com/science/article/pii/S2666154323000662]
- 76. Control of Helicobacter pylori with engineered probiotics ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10580918/]
- 77. Biomarker-responsive engineered probiotic diagnoses ... [https://www.sciencedirect.com/science/article/pii/S1931312822005765]
- 78. Nutritional Factors Affecting Magnesium Bioavailability [https://pubmed.ncbi.nlm.nih.gov/40668512/]
- 79. : clarity in the confusion | WATTPoultry.com Chelates [https://www.wattagnet.com/home/article/15473517/chelates-clarity-in-the-confusion]
- 80. Inorganic Chelated - Bioavailability for Livestock vs Minerals [https://www.wbcil.com/blog/chelated-vs-inorganic-minerals-unlocking-bioavailability-for-peak-livestock-performance/]
- 81. Exploring Mineral Chelates: Mechanisms and Health Benefits [https://www.wbcil.com/blog/understanding-mineral-chelates-mechanisms-and-health-benefits/]
- 82. Impact of Low-Dose Amino Acid-Chelated Trace Minerals ... [https://www.mdpi.com/2076-2615/15/9/1213]
- 83. Chelated Minerals Market Report 2025 [https://www.researchandmarkets.com/reports/5819928/chelated-minerals-market-report?srsltid=AfmBOopZanHeOgZHLdzw5J7s3LwQvV9aBdiHL4djjVoOD2LTLMXFALqW]
- 84. How Chelated Works — In One Simple Flow ( Mineral ) 2025 [https://www.linkedin.com/pulse/how-chelated-mineral-works-one-simple-flow-2025-quantentk-systems-bkbef]
- 85. Drug Formulation Market 2025-2035 [https://www.futuremarketinsights.com/reports/drug-formulation-market]
- 86. 2025 Industry Trends in Formulation Development - USSF [https://www.globalhealthps.com/blog/2025-industry-trends-in-formulation-development-technologies/]
- 87. The Blueprint for Formulation Success Using AI [https://themedicinemaker.com/issues/2025/articles/sept/the-blueprint-for-formulation-success-using-ai/]
- 88. Industry Leaders Weigh in on the Future of Oral Solid Dose [https://www.tabletscapsules.com/3641-Technical-Articles/617375-2025-Predictions-Industry-Leaders-Weigh-in-on-the-Future-of-Oral-Solid-Dose/]
- 89. Flexipill: A novel 3D printed flexible dose combination for ... [https://www.sciencedirect.com/science/article/pii/S0939641125001134]
- 90. Pharmaceutics, Volume 17, Issue 2 (February 2025) [https://www.mdpi.com/1999-4923/17/2]
- 91. 5 Areas Of Drug Delivery Innovation To Watch In 2025 [https://www.drugdeliveryleader.com/doc/areas-of-drug-delivery-innovation-to-watch-in-2025-0001]
- 92. Formulation Optimization and Real-Time Size Monitoring in ... [https://www.sciencedirect.com/science/article/pii/S0928098725003045]
- 93. Biochemistry, Iron Absorption - StatPearls - NCBI Bookshelf [https://www.ncbi.nlm.nih.gov/books/NBK448204/]
- 94. Micro nutrients as immunomodulators in the ageing population [https://immunityageing.biomedcentral.com/articles/10.1186/s12979-024-00492-7]
- 95. Exploring the intricacies of mineral absorption in ... [https://www.sciencedirect.com/science/article/pii/S2590157523005102]
- 96. The Essentials of Vitamin and Mineral Supplementation ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12144039/]
- 97. Multimicronutrient and omega-3 fatty acid supplementation ... [https://www.sciencedirect.com/science/article/pii/S0271531725000818]
- 98. Enhancers of iron absorption: ascorbic acid and other organic acids - PubMed [https://pubmed.ncbi.nlm.nih.gov/15743017/]
- 99. The Best Recovery Supplements for Post-Surgery Patients ... [https://xrscience.org/blogs/education/the-best-recovery-supplements-for-post-surgery-patients-2025-edition]
- 100. Approaches for Enhancing Oral Bioavailability of Peptides and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3680128/]
- 101. Comparative effects of inorganic and three forms of organic trace minerals on growth performance, carcass traits, immunity, and profitability of broilers - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6702935/]
- 102. AG1®, a Novel Synbiotic, Demonstrates Superior Mineral ... [https://www.mdpi.com/2813-0464/2/4/41]
- 103. The role of processing in enhancing mineral bioaccessibility [https://www.sciencedirect.com/science/article/abs/pii/S0960308525001166]
- 104. A review on absorption enhancer and bioenhancer [https://sciencescholar.us/journal/index.php/ijhs/article/view/9908]
- 105. Critical Review on Zeolite Clinoptilolite Safety and Medical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6277462/]
- 106. The Effect of the Combination of Probiotics and Heavy Metals From... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11915010/]
- 107. Penetration Enhancing Agent - an overview [https://www.sciencedirect.com/topics/medicine-and-dentistry/penetration-enhancing-agent]
- 108. Current Development of Chemical Penetration Enhancers ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10044980/]
- 109. Food fortification as a sustainable global strategy to ... [https://link.springer.com/article/10.1007/s44187-025-00512-5]
- 110. Fortified and functional foods: Trends, innovations ... [https://www.sciencedirect.com/science/article/pii/S2666154325006465]
- 111. Detrimental Impacts of Pharmaceutical Excipient PEG400 ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10673170/]
- 112. Advanced Biologics Require Innovative Excipient Science [https://drug-dev.com/special-feature-excipients-advanced-biologics-require-innovative-excipient-science/]
- 113. Drug-Induced Osteoporosis [https://www.uspharmacist.com/article/drug-induced-osteoporosis]
- 114. Emerging Therapeutic Opportunities for Skeletal Restoration [https://pmc.ncbi.nlm.nih.gov/articles/PMC3135105/]
- 115. Medication-induced osteoporosis: screening and treatment ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4206646/]
- 116. Mechanistic Insights and Therapeutic Strategies in ... [https://www.mdpi.com/2227-9059/12/8/1635]
- 117. bisphosphonates and metalloproteinase inhibitors - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC154424/]
- 118. Novel Drug Candidate Could Treat Type 2 Diabetes ... [https://www.pharmacytimes.com/view/novel-drug-candidate-could-treat-type-2-diabetes-osteoporosis]
- 119. Drugs need to be formulated with scale-up in mind [https://www.sciencedirect.com/science/article/pii/S0168365924004474]
- 120. SCALE-UP & MANUFACTURING - Smart Formulation ... [https://drug-dev.com/scale-up-manufacturing-smart-formulation-processing-engineering-solutions-to-solve-drug-product-scale-up-manufacturing-challenges-with-minimum-to-no-regulatory-impact/]
- 121. The Importance of Stability Studies in CMC Compliance [https://synergbiopharma.com/importance-of-stability-studies-cmc-compliance/]
- 122. Hand-in-hand: good manufacturing practice and stability ... [https://www.iptonline.com/articles/hand-in-hand-good-manufacturing-practice-and-stability-studies]
- 123. 4 Factors Influencing the Stability of Medicinal Products [https://www.qbdgroup.com/en/blog/factors-influencing-the-stability-of-medicinal-products]
- 124. Adherence to vitamin and mineral supplementation after ... [https://pubmed.ncbi.nlm.nih.gov/36151032/]
- 125. Adherence to vitamin and mineral supplementation after ... [https://www.sciencedirect.com/science/article/pii/S1871403X22000862]