Research Articles
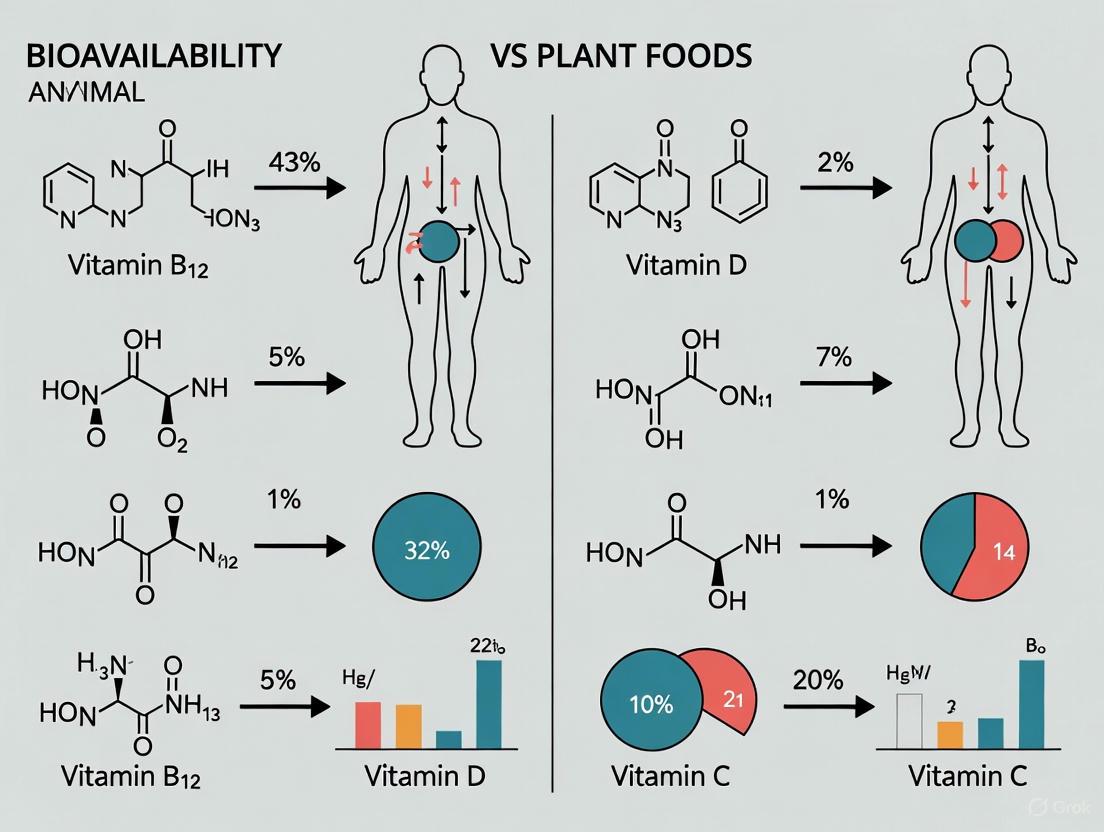
Vitamin Bioavailability in Animal vs. Plant Foods: A Scientific Review for Biomedical Research and Development
This article provides a comprehensive analysis of the bioavailability of vitamins from animal and plant food sources, tailored for researchers, scientists, and drug development professionals.
Predictive Equations for Nutrient Bioavailability: A Framework for Enhanced Nutrition Assessment and Formulation
This article provides a comprehensive overview of the development, application, and validation of predictive equations for estimating nutrient bioavailability.
Nutrient Absorption and Utilization: From Molecular Mechanisms to Clinical Applications in Drug Development
This article provides a comprehensive analysis of the principles governing nutrient absorption and utilization, tailored for researchers, scientists, and drug development professionals.
The Gut Microbiome's Role in Nutrient Bioavailability: Mechanisms, Modulation, and Therapeutic Potential
This article explores the critical role of the gut microbiome in modulating nutrient bioavailability, a key interface between diet, host physiology, and health.
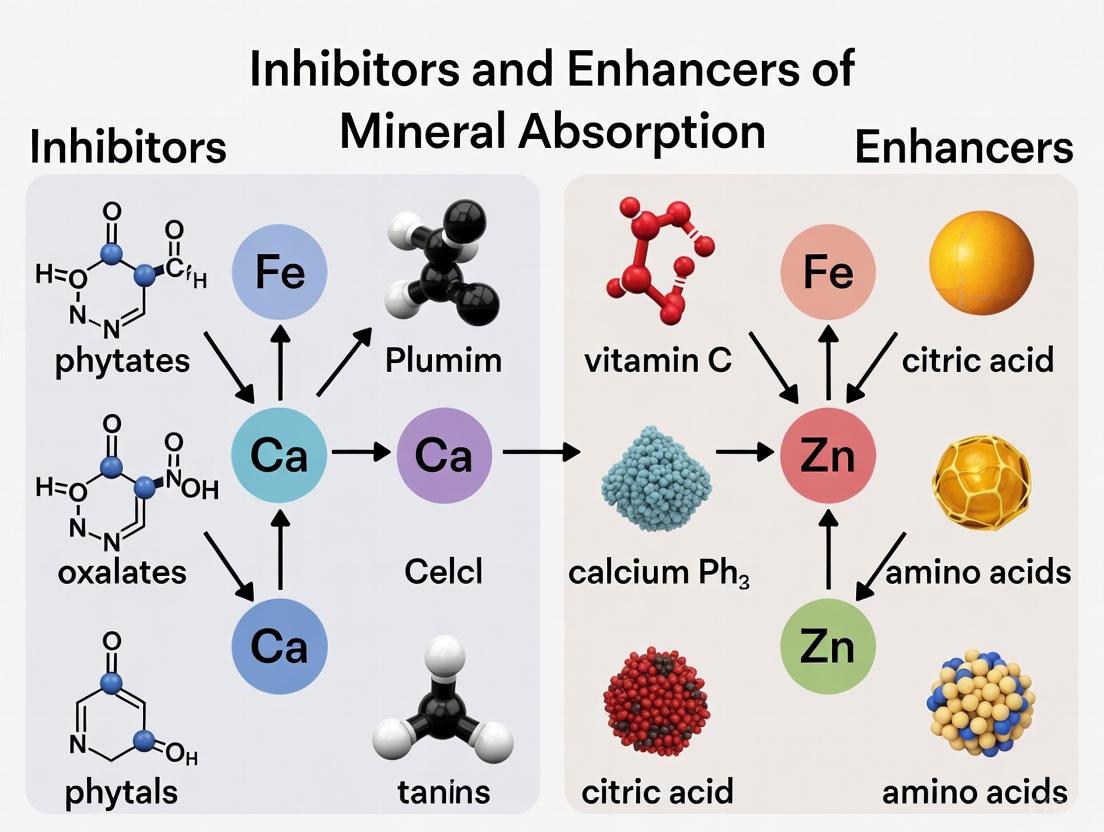
Mineral Absorption Modulators: Mechanisms, Methodologies, and Clinical Translation for Drug Development
This comprehensive review synthesizes current scientific knowledge on the complex factors governing mineral bioavailability, with a specific focus on applications in pharmaceutical and therapeutic development.
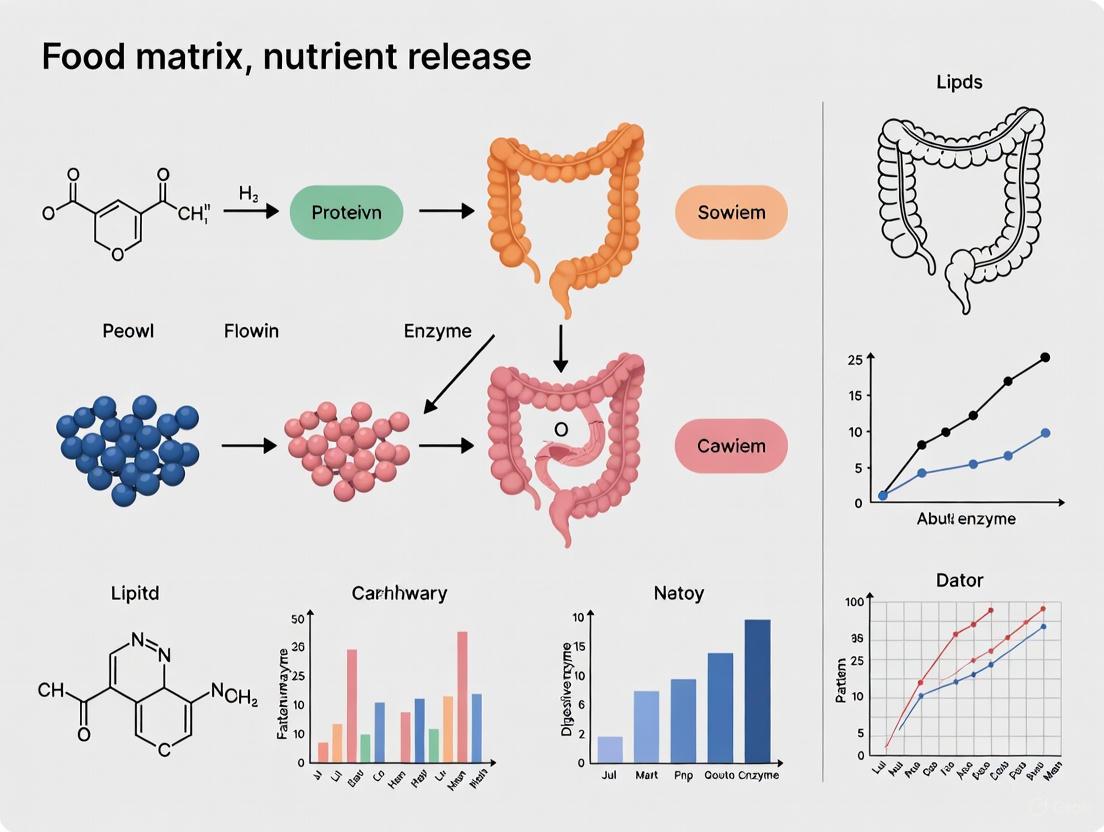
Beyond Nutrient Composition: The Critical Role of Food Matrix in Controlling Nutrient Release and Bioavailability
This article provides a comprehensive analysis of the food matrix and its profound influence on nutrient release, bioavailability, and subsequent physiological effects.
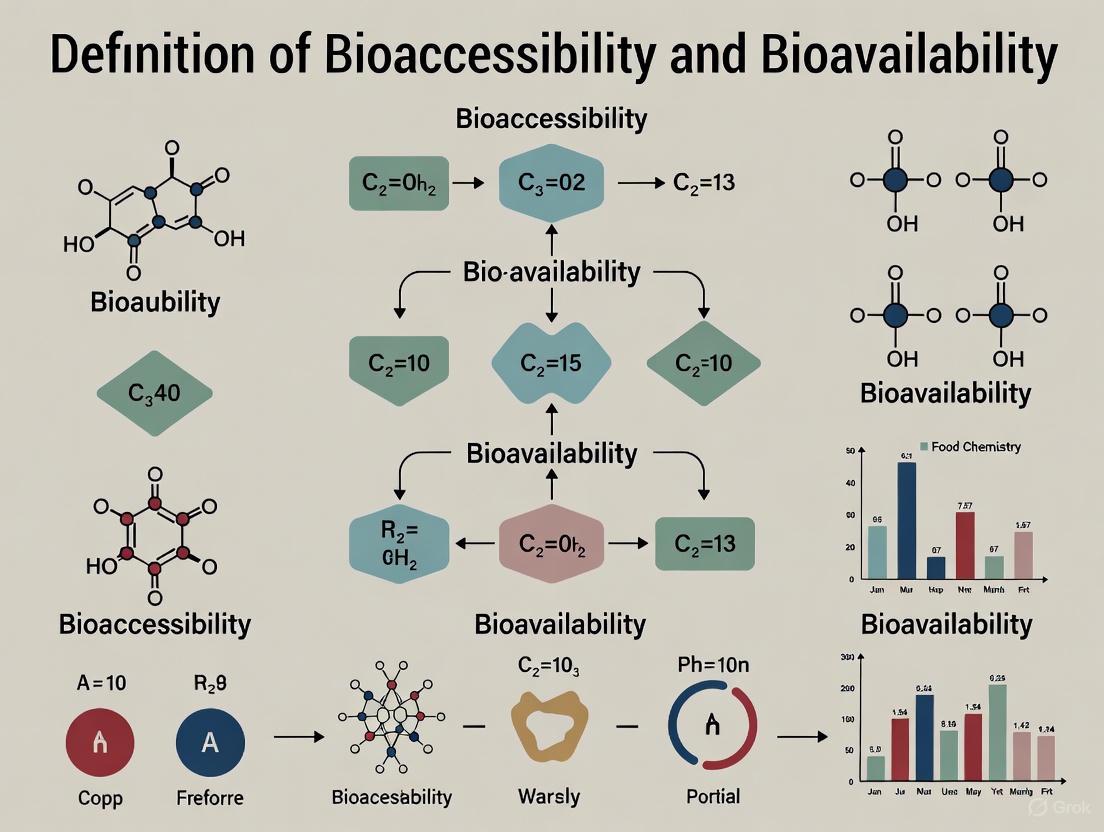
Bioaccessibility vs. Bioavailability: Foundational Concepts, Assessment Methods, and Clinical Relevance for Researchers
This article provides a comprehensive analysis of bioaccessibility and bioavailability, two critical parameters in drug and nutraceutical development.
Decoding Nutrient Bioavailability: Key Dietary, Host, and Methodological Factors for Biomedical Research
This article provides a comprehensive analysis of the multifaceted factors governing nutrient bioavailability, tailored for researchers, scientists, and drug development professionals.
A Strategic Framework for Benchmarking Product Texture in Pharmaceutical Development
This article provides a comprehensive framework for researchers and drug development professionals to benchmark product texture against competitors.
Empirical vs. Fundamental Rheological Tests: A Comparative Guide for Pharmaceutical Researchers
This article provides a comprehensive comparison of empirical and fundamental rheological testing methodologies, tailored for researchers and professionals in drug development.